The Next Generation of Electrical Contacts for Medical Implants
“Medical devices that alter brain, spine, or nerve activity for therapeutic benefit are a fast-growing segment of the healthcare industry,” notes a recent report from Life Science Alley, a Minnesota-based medical device trade association. The 2015 report, Sector Landscapes: Neuromodulation, projects that the strong global market for neuromodulation devices will double by 2018, reaching $6 billion to $7 billion. This article examines the electrical contact technologies that are driving this market toward the next generation of device development.
The global neurostimulation/neuromodulation market is surging as an aging population faces diseases such as Alzheimer’s, epilepsy, spinal-cord injury, and Parkinson’s disease. The therapies for these diseases are dominated by implantable neurostimulation devices, which make up the largest part of this market with a 96% share of the total. According the Life Science Alley report, neuromodulation devices are registered in more than 1,000 ongoing FDA-regulated clinical trials worldwide in which more than 1,300 indications, including metabolic disorders, inflammation, migraine, and psychiatric disorders are being investigated.
The report also notes that one of the key developments within this growing market is the emergence of closed-loop systems that are “capable of sensing ongoing brain or nerve activity and incorporating it into stimulation parameters for optimized therapeutic efficacy in real-time.” This closed-loop functionality in active implantables requires optimal power efficiency and signal isolation, and much of this is dependent upon the performance of electrical contacts and contact systems used to connect leads to batteries and electronics in these devices.
Highly conductive and space-efficient components, such as the Bal Conn electrical contact and the Sygnus implantable contact system, are designed to support the signal-isolation requirements of closed-loop, high-connector-count arrays. With the use of closed-loop devices becoming more widespread, the ability to support smaller footprint connections for active implantables in a proven and reliable design will be increasingly critical.
Unpacking the Technology
For over two decades, the Bal Conn electrical contact has been helping manufacturers improve implantable medical-device performance and push the technology envelope. OEMs developing each generation of active implantables have relied on electrical connections that use canted coil springs as contacts. It’s estimated that canted coil spring–based contacts have been used in more than a million pacemakers, defibrillators, neurostimulators, and other active implantables that deliver life-improving therapies to patients worldwide. These electrical contacts have also dramatically simplified the process surgeons use to connect leads to implantables across the therapy spectrum, including cardiac healthcare, pain management, and sensing therapies, ultimately shortening procedure times.
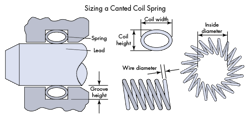
Developed in cooperation with a medical electronics manufacturer seeking to reduce package size while increasing reliability, the Bal Conn electrical contact is an electrically conductive component consisting of a precision-engineered canted coil spring retained in a metal housing. The contact, which is molded into a device header, facilitates a uniform, consistent electrical connection between the lead and the battery in active implantable devices.
The Bal Conn’s individual spring coils provide multipoint conductivity, adjusting individually to maintain maximum contact with electrodes on the lead that is inserted into the device header. The contact’s compact size allows for greater connector density where space is limited, and its unique design eliminates the need for tools during the connection process.
Due to its redundant contact points, the Bal Conn offers low contact resistance, and its canted coils provide excellent resistance to fatigue. With its ability to offer low insertion force and exceptional electrical conductivity, it is ideal for use in devices with high connection counts such as neurostimulators.
In a typical neuromodulation device, the lead diameter ranges from 0.9 to 1.4 mm. The Bal Conn is sized so that its spring coils exert a measured, consistent force, also called the breakout force, on the lead when it is first inserted. The force needed to slide the lead further in, the running force, is as predictable and reliable as the breakout force, and engineers can adjust these forces through design. These forces are low enough to make it easy for surgeons to insert and remove the lead. Applications requiring light forces have a theoretical breakout force as low as 0.5 N and a running force as low as 0.2 N, depending on design parameters. For those applications requiring heavy forces for latching, the theoretical breakout forces are as high as 15 N and the running forces are as high as 4 N with a single spring.
The spring material for neuromodulation applications is typically platinum iridium. Platinum iridium is selected for its biocompatibility, durability, electrical conductivity, and radiopacity. The housing for neuro applications can be made from a variety of materials, including MP35N, 316L stainless steel, medical-grade titanium, or platinum iridium.
Within the contact, both the coils of the spring and the spring itself have precise diameters, but there is no “standard” size for neuromodulation devices. Each Bal Conn is custom designed to fit the particular unit’s lead interface, and the lead is held in place via frictional fit. A set screw or locking detent, located at either end of the channel into which the lead slides, anchors the lead in place. Once the electrodes are aligned with each Bal Conn, the device becomes operational. The spring electrically connects the implantable pulse generator to the lead through all of its individual coils.
Using the spring as an electrical contact provides redundant paths for power and information signals, and the redundant contact points provide for low contact resistance. Their canted coil design helps ensure that they resist compression set and fatigue, providing consistent performance over a long operational life, one that typically exceeds that of the implantable device itself. The springs compensate for imperfections on the surface of leads, accommodating a certain amount of misalignment.
Engineers at Bal Seal Engineering work with medical OEMs to ensure that they have the right design for the application. With its long history of designing for active implantables, the company has developed both uni-directional and bi-directional contacts, which enables OEM integrators to choose which design works best for their particular requirements. With uni-directional versions, the lead must be inserted from one specific side of the contacts. With bi-directional contacts, the lead can be inserted from either side. For neuro devices, the contacts are always bi-directional. Both types are available for other active implantables; however, once integrated into the IPG header, the distinction is no longer significant and has no impact on the surgeon or device functionality.
Simplifying Design for OEMs
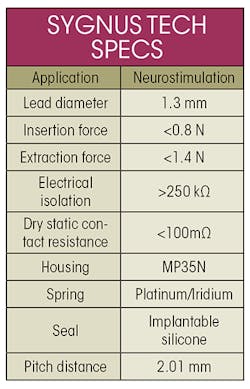
The Sygnus implantable contact system, which combines electrical contacts and isolation seals in a standardized, platform-ready stack configuration, is designed to improve speed to market and eliminate the need for procurement and testing of individual components. It lets OEMs concentrate on the design and function of the implantable rather than worrying about the intricacies and technology behind the connectors. It also creates greater connector density with pitches, or distances between contacts.
Implantable-grade silicone isolation seals keep the contacts electrically separated, as well as separating the different segments of the lead. The seals help prevent signal leakage that can disrupt the active implantable’s function. The result is a densely packed connector stack that accommodates leads with diameters down to 0.7 mm. A 0.64 mm version is currently under development. Sygnus can be modified to suit device-manufacturer requirements for size and number of connections, as long as they are within design limits.
The seal design and dielectric materials were tested in accordance to industry standards by submerging them in saline, a good substitute for the conditions inside the human body. The goal of the testing is to measure impedance between connectors in a simulated body condition. The seals are cleaned to 10K cleanroom standards and packaged in antistatic bags to prevent contamination.
Evergreen Medical Technologies has used Sygnus technology in its device development, including in its Encompass Lead-Interconnect System. In the Encompass, the Sygnus system is combined with a pre-molded 16-channel header. By combining the header, connector, sealing components, and lead into a single, reliable system, the system lets companies developing neurostimulators shorten the time needed to design, develop, source, and test the final device. Although it was originally designed for implanted neurostimulator devices, the system could find use in other implantables as well.
Preparing for the Next Generation
Future developments are being driven by the need for higher connection counts, more compact footprints, closed-loop configurations, and high levels of manufacturability. One reason for such improvements, especially the move toward smaller lead interfaces, is to give OEMs the platform on which to build cutting-edge treatments for hearing and vision loss. One of the engineering challenges they must overcome is that as the connectors get smaller, tolerances become more crucial and manufacturability becomes more complex.
With the use of closed-loop devices becoming more widespread, the ability to support smaller-footprint connections for active implantables in a proven and reliable design will become increasingly critical. To address this, Bal Seal Engineering has developed a new sub-assembly concept to help engineers dramatically improve device functionality while reducing overall package size. The concept, which can incorporate a “cap” or cover design, uses vertically positioned pins or rods of varying diameters to double or triple the amount of connections available to the device lead. It can be engineered to minimize insertion force issues that present challenges in serial arrays with small leads, and it offers designers new opportunities for improved contact density.
Essentially a “build up, not out” approach that challenges the established linear contact/header layout, the high-density vertical array was conceived to help OEMs pack more functionality into active implantables and shrink the size of the electrical connectors in medical devices. The array combines one or more canted coil-spring contacts, a non-conductive polymer interface, and implant-grade silicone. Some of the uses envisioned for the vertical array are in implantables where small size is critical, such as cochlear implants and deep-brain simulators.
Life Science Alley’s report points out that the complex nature of neurodegenerative disorders has set the stage for “numerous potential physiological targets, low competition in treatment-resistant diseases, and significant drive for innovative treatments and novel technology.” As active implantables in neurotechnology continue to advance and prove to be the gold standard for therapy, there will be increased adoption of these life-changing devices. And, as advances such as closed-loop configurations lead to the need for higher connection counts and more compact footprints, design engineers will be faced with the challenge of making the required number of connections in less space with proven underlying technologies.