A Look at the Future of Medical 3D Printing, Part 2
In part one of this series we covered the creation of customized prosthetics, implants, and anatomical models. In part two, the future of 3D printing tissues, organs, and custom pharmaceuticals will be discussed. The idea of printing complex organs is getting a lot of hype, but still sounds like it is a long way off from becoming reality.
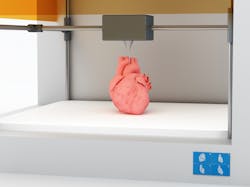
However, an estimate published in the U.S. National Library of Medicine says that we are less than 20 years from a fully functioning printable heart. In addition, 3D bioprinting is also getting hype for the ability to offer exact micro doses of drugs. These advancements have a lot of regulations to pass, but still seem to be moving forward.
3D printing is making its way into many markets, and in medical it is seeping into most of the entire industry.
The More Distant Future of Bioprinting
Currently, bioprinting in place, or in situ, is being employed to repair external organs such as skin. However, being able to print living tissue for implants and complex organs is the ultimate goal. According to the NLM report: “In 2009, 154,324 patients in the U.S. were waiting for an organ. Only 27,996 of them (18%) received an organ transplant, and 8,863 (25 per day) died while on the waiting list. As of early 2014, approximately 120,000 people in the U.S. were awaiting an organ transplant. Organ transplant surgery and follow-up is also expensive, costing more than $300 billion in 2012.
“An additional problem is that organ transplantation involves the often difficult task of finding a donor who is a tissue match,” the report continued. “This problem could likely be eliminated by using cells taken from the organ transplant patient’s own body to build a replacement organ. This would minimize the risk of tissue rejection, as well as the need to take lifelong immunosuppressants.”
The earliest estimate for 3D printed complex organs is less than 20 years.
The report continued by laying out the process of the bioprinting process. This is essentially an inkjet-based process that uses living cells instead of ink, and can build layers in the z-axis. Multiple printheads can be used to deposit different cells exactly where needed. The process aims to:
- Create a blueprint of an organ with its vascular architecture
- Generate a bioprinting process plan
- Isolate stem cells
- Differentiate the stem cells into organ-specific cells
- Prepare bio-ink reservoirs with organ-specific cells, blood vessel cells, and support medium, and then load them into the printer
- Bioprint
- Place the bio-printed organ in a bioreactor prior to transplantation.
Inkjet and laser printers have provided a proof of concept to create a knee meniscus, heart valve, spinal disk, other types of cartilage and bone, and an artificial ear. There has even been an ear created that would extend to frequencies beyond the ability of our natural ears (Thus offering the first proof of concept for a real-life Six Million Dollar Man).
Limitations
Currently, working organs are being printed in miniature. The problem is scaling. The diffusion of oxygen from the host vasculature will nourish the transplanted tissue (often vascular, lymphatic, or nerve cells). Unfortunately, once the engineered tissue exceeds 150–200 micro meters, it no longer supports the oxygen diffusion needed between host and transplanted tissue. This mean to get a complex organ, scientist will need to find a way to scale up with an advanced multicellular structure with vascular network integrated into the part.
So far this has not been accomplished. And once it has, there is a lot of testing and regulations to get through before the technology will be able to be used on a human. Again, all of this is exciting, but as many complex organs are made of thick tissue, patience is called for.
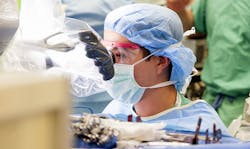
Carlos Villamizar, a medical engineer technician at Walter Reed Army Medical Center, uses a dental pick to scratch between the crevices of a 3D “medical model,” or replica, of a male patient’s lumbar spine. Walter Reed’s 3D Medical Applications Center uses technology originally designed for the manufacturing industry to cut down on the amount of time a patient is in surgery by as much as six hours. (Defense Dept. photo by Fred W. Baker III)
There are issues with the printers, too. In case of trauma, or where speed is important, 3D printers can’t just push out a perfectly matching part or organ in seconds. To perform a scan, transfer it into a CAD file to alter it into what is needed, transfer it into an STL file, and print the organ takes time. In addition, it will take time to print the needed part. Also, the skill and knowledge to take a scan from start to finished organ is not one that any hospital currently has (as the technology doesn’t exist yet).
Furthermore, the cost of the printer might cause a barrier to entry. While it may seem like hospitals have plenty of money, some wouldn’t be able to purchase a high-end printer that might cost hundreds of thousands of dollars. As is the case with many industries, this will lead to printing services being performed by third parties, further delaying any 3D bioprinting part from getting to the patient. These third parties might also have their hands tied by regulations that might further delay organ or part delivery.
Regulations are the main reason for new medical devices to have a long time to market—and for good reason. However, even the FDA started a group to assess technical and regulatory considerations regarding 3D printing. There was also a series of workshop and webinar in 2014 regarding technical considerations of 3D-printed medical devices. Despite this, the FDA still has long and large randomized controlled trials, which require time and funding.
In addition, each state might have its own regulations that will cause further delays. This will be an issue for 3D printing drugs. Often specific doses must go through testing and large, randomized control trails. The idea of 3D printing the exact dose a patient needs could mean testing for each individual pill. Regulations obviously will need to work with the technology but, again, this will take time.
Custom 3D-Printed Dosage Forms and Drug Delivery Devices
Personalized Drug Dosing. A patient’s specific health needs could be mer, and a specific drug or drugs could be printed into a pill for exactly what is needed. For example, if 300mg of a drug is needed, but there is only a pill available in 200 and 500 mg, what would be the right dosage?
Complex Drug-Release Profiles. One of the most researched uses of 3D printing is the complex timed release of drugs. Most drugs are homogeneous compressed doses. Often they are a mix of time- and immediate-release doses. With the ability to print powders in layers, and even the option of printing binders in each layer, 3D printing could offer a concoction of drugs released over time just as the body needs it. People who take multiple pills per day could have them all printed into a single pill. Layers are typically 200 micro meters thick, and with mixing chemicals, adding binders, and barriers, the pharmaceutical industry has a billion-dollar disruptive technology.
This new personalized medicine could even be advanced to where a pharmacy simple carries the basic elements. Then the pharmaceutical manufacturers are replaced with an e-mail from the doctors, a database of medication formulas, and a 3D printer.
A potential problem is that drugs could be 3D printed. This means if this technology is released it might only be a matter of time until someone uses it for criminal activity. Glasgow chemist Lee Cronin found a way to turn a 3D printer into a universal mixing chemistry set. Since most drugs are just mixes of carbon, hydrogen, oxygen, and perhaps a mix of paraffin and vegetable oil, a 3D printer could use these materials to print whatever you like. While the technology seems to be a bit off in the future, this is enough to worry law enforcement.
Ultimately, sciences push on with the idea that printing complex organs and drugs will cause more benefit than harm. However, the same regulations that are delaying this technology will also be the same regulations that keep this technology in the right hands.
About the Author
Jeff Kerns
Technology Editor
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: