Flexible Hybrid Electronics: The Foundation of Tomorrow’s Medical Sensor Devices
In the past 10 years, the consumer healthcare market has seen a dramatic surge in medical devices that were once only available to hospitals and medical professionals. These include pulse oximeters, IR thermometers, automated heart rate monitors, and even active intervention devices such as automated external defibrillators (AEDs). Such devices untether patients and make it possible for doctors to treat patients remotely rather than make them come into a clinic or hospital.
To keep pace with the increasing demand for outpatient monitoring and home diagnostics, engineers have been improving flexible hybrid electronics (FHE) and the data-driven technologies that can be added to wearable devices. They are also developing stretchable interfaces to connect patients and devices, and that improve data collection and the accuracy of that da
Four wireless EKG sensors are ready for assembly. (Photo by GE Global Research)
Collecting patient data at regular intervals and more frequently than, say, an annual or six-month visit, can create a “personal baseline” for patients. For example, a patient’s heart rate or blood pressure as compared to a global average that blends many factors, is not as important as comparing it to what it was this morning, yesterday, last week, and last month. It is this latter comparison that often indicates a need for intervention.
It is telling that we now have better data histories on our automobiles than we have on ourselves. Today, the average length of a doctor visit in the U.S. is less than 20 minutes, and in the UK it is less than eight minutes, according to a study by the Society of Internal Medicine.
In this scenario, the doctor references data points which can be three to 18 months apart, and then bases conclusions and recommendations on a combination of other cases they have seen, their vague memory of your personal history, your anecdotal account, their training, and their instinct. This is not a scientific process. Improved personal baseline data is a good way to improve the accuracy of future diagnoses.
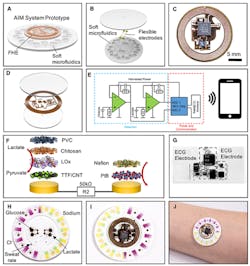
Since NextFlex was formed in 2015, a key focus has been on wearable medical sensors and diagnostic devices. Examples of the organization’s first projects involved development work on individual sensor systems, including oxygen sensing and delivery bandages (led by Purdue University, Integra LifeSciences, and Western Michigan University); electric stimulation sensors and healing devices (led by UC Berkeley and Jabil); a wireless EKG device (led by GE Global Research and Binghamton University); and oral biomarker sensors (led by PARC and UCSD). Each of these sensors incorporates FHE in their design, delivering improved data collection through continuous monitoring.
Today, assembly can take place at below 100˚C, so new sensors can include components that must be kept at relatively low temperatures. This lets manufacturers include materials that would not survive traditional electronics assembly processes that are typically done at around 230˚C. This means biomaterials such as a bacteriophages and certain proteins can be included in the sensor, opening the door to entirely new sensor types.
Flexible circuits with stretchable substrates that conform closely to the patient’s skin improve data transmission and accuracy. In many cases, the ability to conform to various patients also improves the process of applying sensors, which also improves diagnostic accuracy.
These images who how film or sensor thickness affects how well it conforms to the surface of the skin, with the thinnest film conforming the best. (Photo by UT Austin)
In addition to improved conformability, sensors can be smaller and thinner—so they are less obtrusive and bulky, and more comfortable—and therefore more likely to remain in place. Patients are also more likely to wear comfortable sensors, so a continuous data stream from the patient can be established.
Overall, it is the combination of data from an array of wearable sensors and the correlations between them that will yield the real breakthroughs. For example, one medical researcher has shown that looking at heart rate and body temperature predicts exposure to certain types of viruses much more accurately than using either reading on its own.
Companies now are developing multi-sensor wearable devices. For example, one company’s multi-sensor devices measure a patient’s sweat chemistry.
These images show how a sweat sensor is constructed. (Image by Epicore Biosystems)
Another firm is developing a multi-sensor applique with mid-range wireless RF transmission and low power consumption that monitors and records any sensor feeds sent to it.
A third company is combining several clinical-grade medical sensors to eliminate wires and leads on hospital patients with the goal of monitoring patients in the comfort of their homes. In one hospital, nearly 300 care providers each monitor three or more patients from consoles.
This program has been so successful that the hospital has contracted with three other hospital chains to provide this same capability. The hospital has also learned that patients can recover at home and still be well monitored throughout the recovery process. Even better, they also discovered that patients recover faster and costs are lowered.
There are many advantages and benefits FHE offers in terms of healthcare sensor systems. Organizations such as ours are developing not only the sensors, but the fundamental research to prove these devices are possible and can be manufactured in the volumes needed to bring down prices and make the sensors common across the globe.
About the Author
Jason Marsh
Director of Technology
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: