Lab Develops Viscosity Profiles for Carbon-Binding Materials
With the aid of computer modeling, a team from the U.S. Department of Energy’s Pacific Northwest National Laboratory (PNNL) is investigating the behavior of CO2BOLs when binding to CO2 emissions. CO2BOLs, the acronym for carbon-dioxide binding organic liquids, are used to bind emissions and exhaust so that they may be scrubbed. The residual CO2 then can be used for fuel or in other processes.
But as CO2BOLs bind to more and more carbon, viscosity increases to levels whereby they become hard to process and can be harmful to machinery and piping. Thus, PNNL scientists were tasked with finding out which form of CO2BOLs would bind the most carbon dioxide while maintaining a low viscosity.
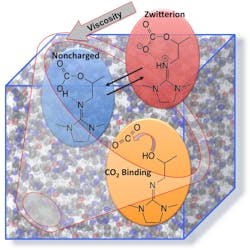
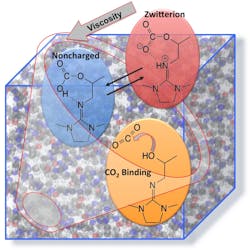
The research, led by chemist Roger Rousseau and his colleague Vassiliki-Alexandra "Vanda" Glezakou, began with investigating a simple CO2BOL called IPADM. Capable of keeping track of the position and movement of thousands of atoms, the computer program enabled the scientists to observe interaction between IPADM with CO2.
The simulations showed that when neutral CO2 bound to IPADM, it generated a IPADM-CO2 molecule with a positive and negative charge on different parts of the molecule. This ion is called a “zwitterion”, which is German for hybrid-ion, because it is a hybrid of a cation and anion. In this particular CO2BOL, the zwitterion IPADM-CO2 is most easily and most abundantly formed.
When binding CO2 to IPADM in a real-life scenario, the scientists realized that the generated zwitterion is the most likely culprit for driving up the viscosity during carbon-dioxide binding. They rationalized this thought by saying that the opposite charges caused interlocking between individual IPADM-CO2 molecules and reduced their ability to move freely.
As a result, the scientists began to simulate carbon-dioxide binding to two other CO2BOLs to see if more quantities of neutral acids would be generated, rather than zwitterions. In one of the CO2BOLs, called EODM, the simulation showed that binding CO2 generated 50% neutral acid and 50% zwitterions. They tested the viscosity of EODM-CO2 in real life and found that it had 50% lower viscosity than IPADM when bound to the same amount of carbon dioxide.
Testing on more CO2BOLs is currently underway, to observe their abilities to generate more neutral acids rather than dipolar zwitterions during carbon binding. By leveraging the simulation software, they hope to develop higher-capacity CO2BOLs that can bind to more CO2 with lower rises in viscosity.
The PNNL published their article in Physical Chemistry Letters.
About the Author
Leah Scully
Associate Content Producer
Leah Scully is a graduate of The College of New Jersey. She has a BS degree in Biomedical Engineering with a mechanical specialization. Leah is responsible for Machine Design’s news items that cover industry trends, research, and applied science and engineering, along with product galleries. Visit her on Facebook, or view her profile on LinkedIn.