Cell-Diamond Anvil Almost Compresses Hydrogen to Metallic Phase
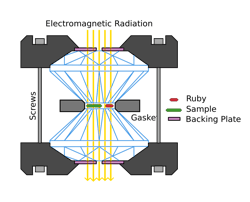
Most often, scientists try to create metallic hydrogen using cell diamond anvils that compress miniscule amounts of hydrogen gas between the tips of high-quality diamonds. Through decades of experimentation, scientists have been able to observe crystallization to some extent, but converting hydrogen, a gaseous insulator, into a metallic conductor of heat and electricity, requires ultra-high pressures that can last long enough to be measured. And since any imperfections in the diamonds can cause them to crack, experiments can be tedious and expensive.
The latest development in creating metallic hydrogen comes from an interdisciplinary team of scientists at the University of Edinburg, Scotland. They used a cell diamond anvil to generate pressures higher than 325 GPa and just below 400 GPa—roughly 3- to 4-million times that of the earth’s atmospheric pressure. This was done at 300 Kelvin, which is approximately room temperature, to produce a phase of hydrogen proceeding the metallic solid phase. Results are published in Nature Letters.
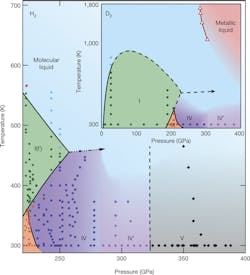
Since only small samples of hydrogen can be compressed for short periods of time, scientists have historically been unable to present results for metallic hydrogen. Still, empirical results and theoretical calculations have enabled scientists to create a phase diagram for hydrogen at high temperatures and pressures (see left).The Edinburg team used Raman spectroscopy to measure phase changes in their samples, which measures vibrational modes of atoms that are characteristic to their quantum state in a lattice. They also conducted their study at room temperature so they could observe a solid, rather than an unstructured liquid.
At these conditions, the team proved that they could turn diatomic hydrogen (H2) and the heavier hydrogen deuterated (HD) isotope into phase V, which is a precursor to the solid metallic state predicted in 1935 (phase VI). When testing a heavier isotope of hydrogen, deuterium (D2), the team was only able to achieve phase IV’. The phases achieved by the Edinburg team can be located on the phase diagram by scanning across at 300 Kelvin. Developments from Edinburg bring us a step closer to documenting such an elusive phase of hydrogen.
But the scientists still need to prove to their community that their results are valid. In 2012, Mikhail Eremets and Ivan Toyan from the the Max Planck Institute for Chemistry in Mainz, Germany went through about 100 diamonds before achieving significant pressures that would give them any results. (Read the full article here.) And like other the experiments that claimed to have created metallic hydrogen, their results were met with reasonable criticism.
There are many potential uses if we are able to controllably create metallic hydrogen. Since 2011, NASA has investigated its uses as rocket fuel. Similar to the way that diamonds do not revert back to coal after they are extracted from the mine, meta-stable metallic hydrogen would remain metallic after it’s removed from high pressure conditions. The energy released by nuclear fission when reverting atomic metallic hydrogen back to a molecular insulator could be used to propel spaceships for lighter payloads and less fuel.
In a paper written in 1935 in the AIP Journal of Chemical Physics, Eugene Wigner and H.B. Huntington predicted that under extremely high pressures, molecular hydrogen would transform from a gaseous insulator into a metallic liquid or solid. This would occur after the hydrogen molecules' diatomic bonds break, producing atomic hydrogen (which is notoriously unstable). The sample would further condense until the atoms form a dense crystal lattice, in which electrons are free to flow between nuclei, carrying a charge at low resistance like they do in a metal. A second study in 1968 predicted that metallic hydrogen at high temperatures would have superconductive properties, where electrons transfer between atoms to carry an electrical charge with zero resistance.
About the Author
Leah Scully
Associate Content Producer
Leah Scully is a graduate of The College of New Jersey. She has a BS degree in Biomedical Engineering with a mechanical specialization. Leah is responsible for Machine Design’s news items that cover industry trends, research, and applied science and engineering, along with product galleries. Visit her on Facebook, or view her profile on LinkedIn.