What's the Difference Between Thorium and Uranium Nuclear Reactors?
The short answer to the question asked above is that uranium-fueled reactors can be built right away, but they use fuel inefficiently. Thorium-fueled reactors, on the other hand, are fuel-efficient, almost perfectly so, but that comes at the end of a three-phase process, with the first phase shared by thorium and uranium fuel.
Starting with Uranium
Uranium is the only element with a naturally occurring fissile isotope, meaning the isotope’s nucleus splits into two smaller nuclei when struck by a neutron traveling at any speed. Few isotopes are fissile. But all thorium and nearly all uranium is fertile, meaning that when they absorb a neutron, they undergo a double-beta decay and become fissile materials.
This has both positive and negative implications.
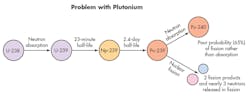
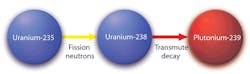
Nearly 100% of natural uranium is the isotope U-238 (99.3% to be exact), so its behavior in a reactor dominates the discussion of nuclear energy. Except in rare circumstances, U-238 won’t fission when it absorbs a neutron, but it does decay on its own over a few days into Pu-239.
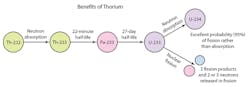
Essentially all natural thorium is the isotope Th-232, and it also does not fission when struck by a neutron (except under rare circumstances). After absorbing a neutron, it decays in about a month to U-233.
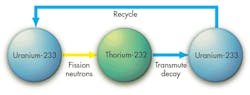
U-233 is special in that it will likely fission when struck by a neutron of any energy and release enough neutrons to continue converting thorium into new U-233. It acts a bit like a “nuclear catalyst,” letting thorium be consumed like a fuel in properly designed thorium reactors.
If a thorium reactor is started with U-233 and it is frugal with its neutrons, the reactor can use thorium as a fuel and release essentially all of its energy. This is the main reason people have gotten excited about thorium reactors. Full energy release means no wasted fuel at the end, which eliminates the problem of long-lived nuclear waste. Of course, such a reactor must be efficient at the chemical recycling step, which is a strong reason why reactors that use liquid fluoride-salt mixtures are so attractive as thorium reactors. But the larger challenge is the need to start the reactor on uranium-233.
We simply don’t have much uranium-233. It does not occur naturally and must be produced from thorium. But it’s difficult for even well-designed thorium reactors to produce much more U-233 than what they consume. A source of U-233 is needed, and that takes us back to uranium and plutonium.
Nuclear engineers have a problem with plutonium-239: it only fissions about 65% of the time it is struck by a slow neutron. The other 35% of the collisions result in absorption and the formation of plutonium-240, which isnât much of a nuclear fuel.
Adding Plutonium
U-235 is the only naturally occurring fissile material, and so it was the starting point for developing nuclear energy. But it is rare: only seven out of 1,000 uranium atoms are U-235.
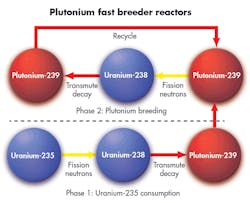
In the earliest reactors, uranium was used at its natural isotopic composition: only 7 atoms out of a thousand. Later, enriching uranium so that the percentage of U-235 is higher let other coolants such as light water be used. But even in these reactors, U-238 outnumbers U-235 by 20 or 30 to one. So as these U-235 atoms fission, they produce neutrons that are absorbed in U-238 and form Pu-239.
But Pu-239 has a problem; it only fissions about 65% of the time it is struck by a slowed-down neutron. The other 35% of the collisions with slowed neutrons result in absorption and the formation of Pu-240, which isn’t much of a nuclear fuel. And therein lies the real problem with uranium-plutonium fuels.
Because of Pu-239’s one-out-of-three absorption problem, it can’t make enough new Pu-239 to replace what was consumed. U-233 in thorium can do it, but Pu-239 in U-238 can’t, at least not with slowed neutrons.
Uranium-233 has a high probability of undergoing fission when it is struck by a neutron of any energy, and fission releases enough neutrons to continue the conversion of thorium to new uranium-233. This lets thorium be consumed like a fuel in properly designed reactors.
This is why today’s reactors have to be regularly shut down to load in fresh fuel and take out some used fuel. They run too low on fuel to keep generating useful nuclear energy. Nuclear engineers call this a “loss of reactivity.”
There are several other neutron-eating culprits in typical reactors: the light hydrogen that likes to absorb neutrons, the metallic cladding of the fuel tubes, and the fission products left over from the reaction. But the central problem is that Pu-239 eats one out of three slowed neutrons instead of fissioning and the reactor steadily runs out of fuel.
For 70 years governments, academics, and nuclear industrialists have championed one possible answer to this problem: When Pu-239 is struck by a fast neutron instead of a slow neutron, the probability it absorbs that neutron instead of fissioning goes way down. And because splitting Pu-239 creates two or three neutrons, there are enough neutrons to make more Pu-239 (from U-238) than was consumed. This is referred to as a “fast breeder reactor.” Remember, the word “fast” has nothing to do with how quickly the reactor makes electricity or how quickly it creates new plutonium fuel. It only refers to the speed of neutrons in the reactor causing fission.
All of today’s reactors intentionally use slow neutrons, and have materials such as water or graphite in their path to slow them down. Such materials need to be eliminated in the reactor if the goal is to ensure a lot of the neutrons are fast. That is why “fast” reactors use coolants such as sodium or lead that are a lot heavier (on an atomic basis) than hydrogen or carbon. This limits the slowing down of neutrons (called neutron moderation) and keeps the neutrons fast, thus raising the likelihood the Pu-239 will fission rather than absorb a neutron.
But fast reactors are challenging machines. The favored coolant for these reactors, metallic liquid sodium, is highly reactive and will react not only with water and air, but with nearly anything that will accept an electron. And we live on a planet full of things ready to accept electrons. Using lead as a coolant solves this problem, but a more fundamental one persists: it is quite challenging to control a reactor whose neutrons are predominantly moving so fast.
Reactors that slow down their neutrons, which is just about every reactor, are straightforward to control because the process of slowing down neutrons builds in a variety of safety mechanisms. In water-cooled reactors, for example, if the water overheats, it expands so it’s less dense and there’s less of it there to slow neutrons, and the fission rate drops, so the water cools. Conversely, if it gets too cool, the water gets denser and the fission rate increases, heating the water. The use of water makes the reactor inherently self-controlling. Fast reactors don’t have this feature because they intentionally don’t have a moderating material such as water or graphite, and that’s because they’re trying to keep Pu-239 from absorbing so many neutrons.
Enter Thorium
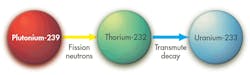
Because plutonium can’t “catalyze” efficient consumption of uranium in reactors with slowed neutrons, and because fast neutron reactors have their own challenges and dangers, perhaps the answer is get the plutonium away from uranium altogether and consume it in the presence of thorium instead. The idea is to remove plutonium, which is chemically distinct from uranium, and let it fission in proximity to thorium. Then the neutrons it emits while fissioning are captured in thorium to create U-233.
This neatly solves several problems. It eliminates the plutonium produced in today’s uranium-fueled reactors and it generates the U-233 needed to efficiently start future thorium-fueled reactors.
A three-phase plan was probably first proposed by Eugene Wigner at the University of Chicago in the fall of 1944. Actually building one has been the goal of India for over 50 years.
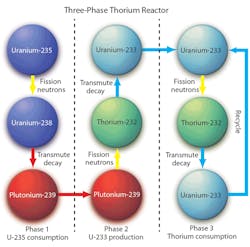
In the three-phase plan for efficiently using thorium, a uranium-fueled reactor consumes U-235 and as it fissions it generate neutrons that are absorbed in U-238 to create Pu-239. The Pu-239 is chemically extracted from the reactor and put into another reactor for the second phase.
The second phase consumes the Pu-239 through fission, releasing neutrons in the presence of thorium that generate U-233, which is also chemically distinct and separable. In the third phase, the U-233 is fissioned and releases neutrons in the presence of thorium, which absorbs the excess neutrons and creates enough U-233 to continue turning out energy (heat) as long as thorium is supplied.
Nearly all of the today’s reactors are representatives of the first phase of this plan. They have been consuming uranium and generating plutonium for many decades now, and that spent nuclear fuel containing plutonium is generally viewed as a hazardous waste, or at the very least, an unappreciated byproduct. Plutonium fissioning in the presence of thorium generates the U-233 that is the bridge, or second phase, between the inefficient first phase and the sustainable, fuel-efficient third stage.
Only one reactor has ever been built that could qualify as a reactor representative of the third phase of the plan, and that was the final core of the Shippingport Atomic Power Station in Pennsylvania. The experiment was called the Light Water Breeder Reactor and involved using thorium/U-233 fuel in a repurposed pressurized water reactor. The experiment was a success and showed that just as much U-233 could be produced as was consumed.
But the pressurized water reactor is not a likely candidate for a sustainable third-phase thorium reactor because the increased potential performance available from thorium can easily be lost on reactors that lose lots of neutrons.
Today’s light-water-cooled reactors lose many neutrons to the light hydrogen in the coolant water that is moderating the neutrons. They also lose neutrons to the fuel rod’s metallic cladding and to the decay byproducts that build up in the solid fuel pellet. If these losses exceed the margin of performance of the thorium fuel, the whole effort to use thorium could be for naught and one would have likely gotten less expensive fuel by just using uranium. The Shippingport experiment succeeded, but chemically recycling the fuel was never attempted.
Reactors that use liquid fluoride salts have far greater potential both for Phase 2 and 3. A particular mixture of fluoride salts, one with highly depleted lithium fluoride and beryllium fluoride, can serve as a suitable solvent while absorbing few neutrons. Using graphite (carbon) as the moderator would involve more material to slow neutrons, but would not absorb them nearly so much as light hydrogen. Graphite is also chemically compatible with fluoride salts, so metallic cladding can be eliminated along with its neutron losses. Thorium, uranium, and plutonium all form suitable fluoride salts that readily dissolve in the LiF-BeF2 mixture, and thorium and uranium can be easily separated from one another in fluoride form, an enormous improvement over today’s ceramic (oxide) fuels.
So what’s the difference between uranium/plutonium and thorium-powered reactors? Uranium-fueled reactors are the first step in any attempt to use nuclear energy because only uranium has an isotope that fissions. But releasing all the energy of uranium is difficult and requires that uranium be transformed to plutonium and consumed through fission in a reactor with fast neutrons.
Thorium-fueled reactors are fuel-efficient as long as they operate using U-233 and chemically process their fuel at high efficiencies, a task liquid-fluoride reactors can handle. The need for U-233 persists, however, and splitting plutonium in liquid-fueled reactors containing thorium is the bridge between the two, permanently eliminating the plutonium from the first phase of nuclear energy while generating the U-233 needed for the next phase. In this way, the full potential of thorium can be harvested, and this would go a long way to improving the public acceptance of thorium technology.
About the Author
Kirk Sorensen
Chief Technologist
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: