Experiments at SLAC’s X-ray laser reveal in atomic detail how two distinct liquid phases in some materials enable fast switching between glassy and crystalline states to represent 0s and 1s in computer memory devices.
Instead of flash drives, the latest generation of smartphones uses materials that change physical states, or phases, to store and retrieve data faster, in less space, and with more energy efficiency. When hit with a pulse of electricity or optical light, these materials switch between glassy and crystalline states that represent the 0s and 1s of the binary code used to store information.
Now scientists have discovered how those phase changes occur on an atomic level. Researchers from DoE’s SLAC National Accelerator Laboratory, the European XFEL, and the University of Duisburg-Essen in Germany conducted X-ray laser experiments at SLAC. They collected more than 10,000 snapshots of phase-change materials transforming from a glassy to a crystalline state in real time.
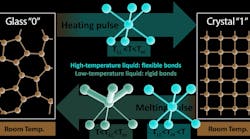
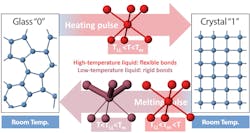
In phase-change memory devices, a material switches between glassy and crystalline phases that represent the 0s and 1s used to store information. One pulse of electricity or light heats the material, causing it to crystallize, and another pulse melts it into a disordered, glassy state. Experiments at SLAC’s X-ray laser revealed a key part of this switch is a quick transition from one liquid-like state to another that enables fast and reliable data storage.
The researchers discovered that just before the material crystallizes, it changes from one liquid-like state to another, a process that could not be clearly seen in prior studies because it was blurred by the atoms’ rapid motions. The photos showed that this transition is responsible for the material’s ability to store information for long periods of time while also quickly switching between states.
The results offer a new strategy for designing better phase-change materials for specialized memory storage.
“Current data storage technology has reached a scaling limit, so that new concepts are required to store the amounts of data we will generate in the future,” says Peter Zalden, European XFEL scientist. “Our study explains how the switching process in a promising new technology can be fast and reliable at the same time.”
The experiments took place at SLAC’s Linac Coherent Light Source (LCLS) which produces X-ray laser pulses short and intense enough to capture images of atomic changes occurring in femtoseconds (millionths of a billionths of a second).
To store information with phase-change materials, they must be cooled quickly to enter a glassy state without crystallizing and remain in this glassy state as long as the information needs to remain there. This means crystallization must be slow to the point of being almost absent, such as is the case in ordinary glass. But when it comes time to erase the information, although it is done by applying high temperatures, the same material has to crystallize quickly. The fact a material can form a stable glass but then become unstable at elevated temperatures has puzzled researchers for decades.
The research team after performing experiments at SLAC’s Linac Coherent Light Source X-ray laser. (Credit: Klaus Sokolowski-Tinte)
At LCLS, scientists used an optical laser to rapidly heat amorphous films of phase-change materials, just 50 nanometers thick, atop an equally thin support. The films cooled into a crystalline state as heat from the laser blast dissipated into the surrounding support structure in just billionths of a second.
They used X-ray laser pulses to make images of the material’s structural evolution, collecting each snapshot the instant before a sample deteriorated.
Researchers found that when the liquid cools far enough below the material’s melting temperature, it structurally changes into a lower-temperature liquid that exists for just billionths of a second.
The two liquids not only have different atomic structures, but also behave differently: The one at higher temperature has highly mobile atoms that can quickly arrange themselves into the well-ordered structure of a crystal. But in the lower-temperature liquid, some chemical bonds become stronger and more rigid and can hold the disordered atomic structure of the glass in place. It is only the rigid nature of these chemical bonds that keeps the glass from crystallizing and, in the case of phase-change memory devices, holds information in place. The results also help scientists understand how other classes of materials form a glass.