Implementing Multiphysics Modeling in the Biopharma Industry
Amgen, a leading multinational biopharmaceutical company, uses multiphysics simulation as a tool in its arsenal at any point in their drug production processes to ensure drug efficacy and safety. The company’s products have treated serious illnesses in millions of people around the world.
But behind every product is a plethora of processes, and Amgen employs a diverse portfolio of process models to enhance its workflow. In an industry where process modeling is more prevalent than product modeling, a portfolio such as this is key. Pablo Rolandi, director of process development at Amgen, has overseen the use of the COMSOL Multiphysics software as a platform modeling tool for his workforce. As various problems in the development phase arise, Rolandi and his team turn to multiphysics modeling as a solution.
In many cases, these solutions are also accompanied by the development of simulation apps, which can be created directly from the model via the Application Builder. By operating a specialized user interface, the end user can still benefit from the insights provided by simulation results, even if they are not experts in modeling. For the last year-and-a-half, the company has developed app packages that are streamlined, communicable, and easily deployed to serve corporate functions in process development, operations, and R&D.
Eliminating Bottlenecks in Production
The optimization of a drying process serves as a great first example where Rolandi’s team developed a custom app to help solve a production workflow issue. This case centered on relocating the manufacturing process of a small molecule drug substance from a contract manufacturing organization (CMO) to Amgen’s plant in Singapore.
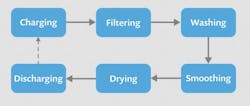
In the midst of this, drying operations—isolations performed by an agitated filter dryer (AFD) in a process similar to the one shown in Fig. 1—were identified as potential bottlenecks in the production facility. Naturally, a bottleneck can pose a substantial risk to meeting product demand, so Rolandi and his team set out to model the drying operations and streamline the process.
Because the CMO used a different type of dryer for the rest three steps of the process, shown below in Fig. 1, it lacked sufficient characterization data from these isolations to accurately model it and identify the impact of changing operating conditions.
1. Shown are basic steps in a typical batch filtration and drying process for the isolation, or physical separation, of a chemical substance.
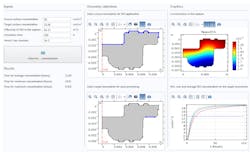
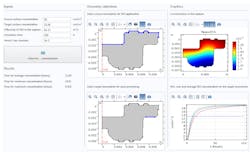
Known properties of the system included material properties, geometric properties of the equipment, and operating conditions—including moisture content, temperature, pressure, and whether agitation is involved. Rolandi, however, still needed to determine two critical factors: the evaporation rate and the diffusion coefficients of the new AFD. To accomplish this, extensive data acquisition was performed and, using multiphysics simulation, his team manually estimated the regressed parameters to characterize the model.
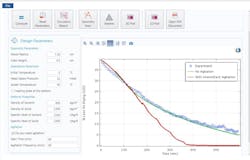
Once this was completed, a simulation app that calculates drying times was created and deployed to process engineers, changing production sites in the pivotal phase. This played a significant role in giving end users the opportunity to visualize the impact of altered operating conditions, as shown in Fig. 2. It was ultimately discovered that the combination of agitation with a heating plate reduced the time to dry, thus helping mitigate the bottleneck and increasing efficiency.
2. This simulation app calculates drying times for models with no agitation (green) and intermittent agitation (red) and compares them with an experimental result.
Ensuring Sterilization Standards Are Met
In another situation, one of Amgen’s production teams encountered an issue with sterilization. Compounds from manufacturers are transported in primary containers. These are often vials and must be sterilized to a certain standard to be classified as a novel container, as bacteria in drug products can pose tremendous health risks. However, the standard sterilization protocol, which involves diffusion of ethylene oxide as the main transport mechanism, was not meeting the requirements for a novel container.
Naturally, the sterilization process needed to be tweaked. But rather than undertake undue experimentation and costly iterations of trial and error, Rolandi and his team took to simulation to model the ethylene oxide being diffused through the vials.
3. This sterilization process app calculates the concentration of ethylene oxide.
The app featured options to select permeation and contamination boundaries and input solubility and diffusivity constants, and generated time-dependent concentration profiles of the ethylene oxide (Fig. 3). Process engineers could then use the apps to determine whether concentration levels were high enough to warrant sufficient sterilization. As a result, experimentation was either reduced or avoided altogether and the program was accelerated by a number of months. “In the end it was much more efficient to just create simulation apps,” Rolandi said.
Beyond Simulation
“I’m very keen on thinking beyond simulation, about the development and integration of very advanced applications and techniques,” Rolandi explained. “I think there is a strategic challenge with that and we are just getting started.” One of his goals is to incorporate uncertainty into the company’s models. In practice, parameters are rarely exact and operating conditions are variable; integrating these variations into their simulations can lead to more predictive results that can be better understood in context.
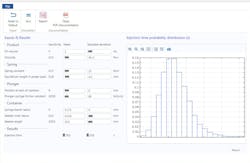
For example, Rolandi and his team are working on an autoinjector, a device that injects medicine into a patient without a physician having to administer it. A critical aspect of injections is the time of delivery; this needs to be controlled very precisely in order for the administered drug to perform as intended. The issue is that the delivery time depends on a number of factors, all with varying degrees of uncertainty. These include the container geometry, the viscosity and volume of the drug, the spring constants of the injector, and the friction constants of the plunger.
If the uncertainty inherent to these factors is not accounted for, a simulated time of delivery will have an unknown variance, thus giving no information on its potential to be precisely controlled. In process modeling, it is invaluable to create a probability distribution of the expected outcomes in order to better understand how the system will behave.
To gain a better grasp of how the uncertainty of these parameters propagates, Rolandi and his team used multiphysics simulation to run a global sensitivity analysis on the system and rigorously quantify the effect of factor variability. The analysis determines a sensitivity index for each parameter, which is a fractional attribution of the variance in response to that parameter.
What the team found was that the viscosity of the product, the spring constants, and the needle geometry accounted for 90% of the variance in injection time, allowing it to greatly simplify its model. Because only a few parameters have significant impacts on the injection time, it is much easier for it to manage uncertainty and risk through robust specifications to component providers.
4. This autoinjector model simulation app shows an injection time probability distribution. Proprietary data have been hidden.
Similar to the company’s other solutions, the injection time model was packaged into a user-friendly and easily-deployed simulation app. The app, shown below in Fig. 4, features user-defined input distributions, runs an uncertainty and sensitivity analysis, writes an automated report, and displays model documentation. The app has delivered cost savings and speed gains and fostered more effective management of uncertainty throughout the entire process.
App Deployment
5. Amgen has personally branded its COMSOL Server Library, naming the system COSMOS.
Amgen also takes advantage of a local installation of the COMSOL Server software to increase accessibility for its employees. “We have an array of applications that we really want to deploy to everybody at Amgen,” Rolandi says. “At the moment, there are about a dozen applications and those are being used today across the organization in a way that I am quite proud of.” With the server, app deployment is trivial, and the company can more effectively manage lifecycles.
Users can simply log in via a web browser to access the application library developed by Rolandi’s team. Amgen also has plans to increase the sophistication within its system by moving away from manual entry and thinking of models as “compute kernels.” These can be repurposed with the help of advanced algorithms in several high-impact, model-based studies, which would mark a major step in implementing enterprise-level modeling that delivers true business value to a large user base and many stakeholders.