How FDA Rules Govern Medical Packaging
Though it is widely understood that medical devices are developed under rigorous quality controls, it is less widely known that the packages that house them are also subjected to rigorous validation processes. The packaging used to seal and deliver medical devices is tested to ensure it will protect the sterility of instruments and implants. The resilient packaging must also meet rigorous labeling standards which let the FDA trace devices in use.
In short, the medical device development is not limited to the devices themselves, but also extends to their packaging and labeling. To learn more about how FDA and industry treats medical packaging, we talked with Jason Haider, founder and CEO of Xenco Medical, a company that makes spinal implants and associated hardware.
Please briefly explain the motivation behind packaging validation.
Jason Haider: Because the FDA has rigorous standards for the sterility of medical devices it approves, manufacturers must undergo thorough testing to demonstrate a package’s resistance to environmental factors that might threaten the sterility of its contents. This testing protects against factors such as the transport and aging of the packaging material itself, which could weaken over time and expose the contents to pathogens.
How does a manufacturer prove packaging is up to standard?
JH: Proving packaging is acceptable package rests on demonstrating the resiliency of the sterile barrier. The sterile barrier prevents entry of pathogens into the package and helps eliminate the risk of non-sterile medical devices making it into surgery in an outpatient surgery center or hospital. The FDA expects its manufacturers to put its packaging through real-time aging test to evaluate how well it handles environmental conditions and preserves the sterility of its contents.
They can also use accelerated aging to demonstrate the sterile barrier’s resiliency. One of the benefits of accelerated aging, though it is more expensive than real-time aging, is that it allows manufacturers launch products earlier and recoup earlier R&D investments faster. Accelerated aging allows manufacturers to capitalize on earlier commercialization of their products.
How does accelerated aging work?
JH: Accelerated aging for validating packaging relies heavily on the Arrhenius equation, which finds that the chemical reactions with activation energies of about 50 kJ per mole, the rate of reaction doubles for every 10°C increase in temperature. With this activation energy, which is the minimum amount of energy required by the objects for a chemical reaction to occur, every 10°C increase above room temperature doubles the rate of molecular collisions occurring in the sample, which results in the material’s degradation and accelerates the aging process.
Manufacturers then use this accelerated aging data to extrapolate the shelf life of the samples at room temperature. Accelerated aging includes the simulation of other conditions, such as humidity, as well.
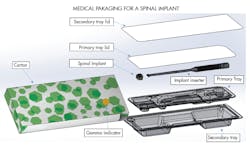
A spinal implant from Xenco Medical and the hardware needed to implant it are shown with its sterile packaging. Note the dual barriers and trays which provide redundant durability and protection against dirt and pathogens.
Are several components in each package measured?
JH: Absolutely. Because the FDA expects a durable sterile barrier, many medical device manufacturers incorporate primary and secondary packaging to ensure durability. In these cases, which are the majority, manufacturers must validate both layers of packaging. Packaging must undergo a spectrum of validating tests to demonstrate their ability to withstand environmental factors. These tests include bubble-emission and burst testing, among others.
In bubble-emission tests, for instance, packages are immersed in water to check for any damages that could compromise the sterile barrier. Any bubbles in the water indicates a path airborne pathogens can follow to get to the contents. In burst tests, air is pumped into a package to determine the pressure at which it will burst.
How is sealing evaluated?
JH: To validate the sealing process, manufacturers must prove the consistency and effectiveness of their sealing equipment. The three parameters generally examined are temperature output of the sealer, dwell time, and input pressure. Validating the temperature output of the sealing equipment includes validating the accuracy of the temperature controller and the consistency of temperature on the heating bars—the components that melt the sealant layers to each other to close the package.
Dwell time is validated by demonstrating that the sealing equipment is in contact with the packaging long enough to consistently seal them. Input pressure is validated by demonstrating there’s enough pressure holding the package components together to effectively and consistently seal them. This ensures the sealing cycle’s reliability, which is the automated process that begins with bringing the packaging materials together and ends with a sealed package.
What qualifying steps does the validation process require?
JH: In the validation process, packaging equipment must pass installation, operational, and performance qualifications. Installation qualification ensures the packaging equipment works. Operational qualification evaluates the processes used to assemble the package. It may also look at aspects such as package sealing. Performance qualification determines whether the packaging processes consistently meet the requirements for a sterile barrier. Essentially, the manufacturer must prove its packaging processes perform as they were intended.
The full packaging on an Anterior Cervical Spine Implant from Xenco Medical shows how the implant is kept sterile during transport and storage.
What is UDI, how does it work, and why has it become so important?
JH: UDI, or unique device identification, has become increasingly important in how manufacturers design and execute their packaging. FDA enforcement of UDI is behind many of the critical decisions medical device manufacturers make on package labeling. UDI is both an identifying link to the global UDI database (GUDID), as well as a direct source of information. GUDID is a database managed by the FDA that stores important identification information on every listed medical device. Manufacturers are required to place a UDI on every medical device label, except in very rare exceptions.
What components make up a UDI?
JH: A key component of the UDI is its device identifier, or DI. It is unique to each model of the device. For example, if a model is updated, the corresponding DI must also be updated. In short, the DI identifies the labeler and the version of the medical device. A separate and less-critical component of the UDI is the production identifier, or PI. Unlike DIs, PIs are not mandatory. They include a device’s lot or batch number, serial number, expiration date, and manufacturing date. If a medical device manufacturer uses API, it must ensure the PI appears in a readable format and a format decipherable by machines. Examples of machine readable formats include linear barcodes and RFID tags. Manufacturers must also follow a prescribed format (YYYY-MM-DD) for putting information about dates on the label.
How do manufacturers know if a UDI has already been used by another manufacturer?
JH: It is difficult for manufacturers to determine if a DI is truly unique, so they are required to work with an accredited agency. These agencies assign unique numbers to manufacturers. UDIs let the FDA improve the traceability and transparency of every medical device through a system that links back to the GUDID.
Though manufacturers may include information such as expiration and manufacturing dates on their labels, the lion’s share of the device’s information is stored in the GUDID, which can be searched using DIs. That is why DIs are mandated. In mandatory submissions of data to the GUDID, manufacturers are required to submit the name of the accredited agency that gave the device its DI.
What about different packaging for same device?
JH: DIs are assigned to every packaging configuration, as well. Manufacturers are also required to say whether a prescription is mandatory for that specific medical device. The DIs let the FDA maintain traceable records of adverse events, and the FDA requires that reports of adverse events include the DIs.
What role does sterilization play in developing medical devices?
JH: It goes without saying that sterilization is an important aspect of product development for most medical devices. For sterilization, several variables are taken into consideration to ensure compliance with FDA expectations. Though there are several ways of sterilizing medical devices, not all are supported by precedents of earlier FDA clearances and a large body of scientific literature.
Category A methods have a long body of supporting history and include ethylene oxide, dry heat, steam sterilization, and radiation. Because these methods have been used in an overwhelming number of medical devices, their effects are largely understood. Gamma sterilization, in particular, has become increasingly popular. Category B methods, on the other hand, do not have FDA-recognized consensus standards. They do, however, have published information on their validation, development, and controls. These refer to sterilization methods such as the use of hydrogen peroxide or ozone.
Dual barriers can also be provided by thermoformed trays, like in this sterile packaging designed for Xenco Medical’s spinal implants.
What aspects of gamma sterilization have made it popular?
JH: Gamma sterilization is particularly well-suited for medical devices that are single-use and packaged after sterilization. Though gamma sterilization is effective, its use depends on the packaging material’s ability to withstand that form of radiation without being damaged. Gamma radiation uses cobalt 60, a radioactive isotope capable of penetrating sealed packages and killing microorganisms. This means gamma radiation can be effective even when a device is in its final package.
How can manufacturers confirm gamma irradiation has been successful?
JH: Gamma radiation indicators placed on packaging can confirm that it has (or has not) been exposed to a sterilizing dose of radiation. After sterilization, audits on each product families ensure radiation levels are under control and sterilization efforts are effective.
Must certain devices be cleaned before sterilization?
JH: A manufacturer may use an ultrasonic bath to clean its medical devices before packaging them. Ultrasonic cleaning reduces the amount of biological material on the device (bioburden). After the cleaned devices are in their packaging, they can be exposed to gamma sterilization, which penetrates through all primary and secondary packaging layers to complete the cleaning and sterilization process.
What is taken into consideration in terms of packaging materials?
JH: When assessing which materials are best for packaging, manufacturers consider their intended function as well as their sterilization characteristics. Rigid thermoformed trays are especially popular among surgical device manufacturers. The materials that make up these trays range from PETG to polycarbonate to HIPS, among others. Packages made from PETG, for instance, are ideal for gamma sterilization. This lets manufacturers streamline development by using a seamless sterilization technology that permeates through the packaging. In contrast to other methods such as steam sterilization or ethylene oxide sterilization, gamma sterilization can sterilize devices in their final packaging, eliminating the need for an additional post-sterilization packaging step after sterilization. Thermoplastic polyurethane films, on the other hand, are used commonly as pouches to hold implants. These pouches are effective as sterile barriers, even though they differ from thermoformed trays. These two types of packaging demonstrate that manufacturers have several different ways to meet FDA expectations for sterile barriers.
How do manufacturers deal with reusable devices after it leaves its original packaging?
JH: The FDA has different expectations for reusable devices designed to be separated from their initial packaging throughout their lifespan (after delivery, of course). As a result, the FDA created guidelines to ensure they are still traceable even though they do not have identifying packaging. In these cases, the FDA expects that devices will have a direct marking traceable to the GUDID. The marking must withstand hospital processing, and cleaning, as well as regular use. The marking may be etched, laser marked, have a permanent plaque, or one of the other methods accepted by the FDA.
How are these markings tracked?
JH: Manufacturers are expected to submit direct markings to the GUDID. If the device will only be reused on the same patient, it does not need to be marked. And if direct marking could degrade the device’s effectiveness or safety, the FDA lets it forgo marking.