Discussing medical technology can be a challenge because it covers a lot of ground these days. The advent of IoT has pushed the majority of devices into this space, so connectivity and security are now even more important. However, much of this shares technology used for more generic IoT devices. In addition, developers need to be aware of certification and safety in the medical space—and, of course, the latest sensor technology makes a difference as well.
Today’s mix of articles touches on many of these aspects from new wire technology for body sensors to
1. BioWire uses recyclable construction consisting of polyethylene terephthalate (PET) and a printed conductive silver ink.
“Nikomed Develops Recyclable Lead Wire from Medical Device and Diagnostic Industry” examines Nikomed’s non-metal disposable and universal lead wire system, BioWire. Thanks to a construction of polyethylene terephthalate (PET) and a printed conductive silver ink, the lead could be recycled.
“The idea for a recyclable lead came from seeing the extremely high volume of inexpensive disposable metal lead wires being thrown away by hospitals,” Stephen Epstein, Nikomed’s president, told MD+DI. “In order to help fight cross contamination, hospitals began looking for alternatives to cleaning/reprocessing traditional, reusable lead wires and turned to inexpensive metal wires from China. These wires are very well made and cost substantially less than reusables. This, too, can be an issue for hospitals and, in turn, patients—many times these ‘disposable’ lead wires are re-used without any reprocessing simply because they look good.”
2. “Robots Are Invading America’s Operating Rooms” examines companies, investments, market size, and types of procedures robotic surgeries are driving.
This article from Machine Design delves into the technology and market driving remote operating-room operations. It takes a look at UC Health’s daVinci Surgical System. The A patient cart has four interactive robotic arms that are controlled from the surgeon’s console. It has been around since 2000.
3. Maxim Integrated’s latest sensors are highlighted in Wearable Clinical-Grade Medical Sensors Get Even Smaller.
Electronic Design’s “Wearable Clinical-Grade Medical Sensors Get Even Smaller” presents Maxim Integrated’s latest, compact clinical-grade medical sensors. These target applications like wireless earbuds. Among the company’s sensors are the MAX86161 in-ear heart-rate monitor and its MAX30208 digital temperature sensor.
4. Wired magazine’s “A Model Hospital Where the Devices Get Hacked—on Purpose” highlights a Defcon Medical Device Village event.
Wired magazine is one I read regularly. Its “A Model Hospital Where the Devices Get Hacked—on Purpose” was generated from Defcon, the annual hackers convention in Las Vegas. The Defcon Medical Device Village event lets hackers target a hospital, albeit a honeypot. “Until very recently, most of this activity would have been not just infeasible, but illegal.”
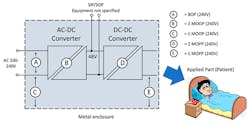
5. This Digi-Key application note highlights how medical device safety ratings can be achieved with the aid of convenient board-mount isolated dc-dc converters.
“Board-Mount DC/DC Converters in Medical Applications” from Digi-Key takes a look at medical devices placed into the market in the European Union. These must meet the essential requirements of directive MDD:93/42/EEC, Article 3, as well as risk-analysis essential requirements set out in Article 3, Annex I, ER1, Annex II, Annex III, and Annex VII. The regulations are to ensure device safety and basic performance; specifically, protection of patients and operators against electric shock. Compliance is presumed if a product conforms to ES/EN/IEC 60601-1, currently in its 4th edition.
About the Author
William G. Wong
Bill Wong is senior content director for Electronic Design.
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: