Advancements in Prosthetic Control Systems
A 2022 study published in the National Library of Medicine at the National Center for Biotechnology Information found that more than 44% of people with upper limb loss abandon their prostheses, citing problems with discomfort, heaviness and functionality. Recently the FDA granted Breakthrough Device Designation and TAP Enrollment to the RESTORE neuromuscular interface from Blue Arbor Technologies.
The startup began last year as a spin-out company from the University of Michigan, where Alex Vaskov, a founder and chief technology officer at Blue Arbor Technologies, earned his Ph.D. in robotics, researching and developing the technology for controlling upper limb prosthetics.
“Since then, we’ve been raising our seed money, and we’ve already started working on building our first device that should be ready for market or clinical trials in 2025,” he told Machine Design.
Vaskov’s doctoral and post-doctoral research focused on the development and clinical testing of real-time control algorithms to interpret peripheral nerve and muscle signals into prosthetic movement commands. Prior to graduate school, he was a software development consultant and had achieved his bachelor’s degree in mechanical engineering from the Massachusetts Institute of Technology.
Key Components of Novel Prosthetic Control Systems
Today’s robotic prostheses exist with great capabilities. Traditional control systems, however, use skin surface electrodes that do not allow the prostheses to function optimally due to their connection.
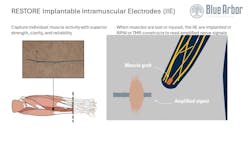
“Signals are immune to environmental disturbances,” Vaskov explains. “So, with surface electrodes…shifts, sweat, loss of contact can all change the control signals. Since the RESTORE electrodes are secured directly to muscles, that makes the control inherently more repeatable and reliable.”
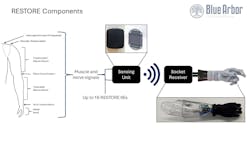
The system, which is designed to be compatible with any commercially available robotic upper-extremity prothesis, is composed of:
- Implantable intramuscular electrodes
- A sensing unit
- A socket-mounted receiver
- A software package.
“[Because] the electrodes are implanted into individual muscles and nerves, we’re getting much higher signal resolution,” Vaskov said. This is what will give the system the capability to let people control individual fingers as well as multiple wrist and hand functions at the same time. “And that’s what’s going to enable people to really fully utilize their prosthetic hands, which isn’t really possible with current control interfaces,” he said.
In addition to the implanted electrodes, which obtain the high-quality signals from muscles, the system’s sensing unit filters, conditions and processes patient-generated electromyographic (EMG) signals and wirelessly transmits these signals to a socket-mounted receiver that decodes these signals into movement commands for the prosthesis.
The sensing unit has up to 16 EMG electrodes connected to it, using standard sampling, filtering, feature and extraction parameters that are commonly used for EMG and that are chosen to optimize for low power consumption and longer battery life, Vaskov explained.
“I think the beauty of [this] system is that we can implant the electrodes into muscles to record EMG from remaining muscles,” he said. “If people are missing muscles because of their amputation, the electrodes can be implanted into a regenerative peripheral nerve interface (RPNI) or targeted muscle reinnervation (TMR).” These surgeries are commonly performed to route nerves to muscle tissue to reconnect the nerve to a body for it to grow into, he says, which has the effect of biologically amplifying motor action potentials.
Because Blue Arbor wants its socket-mounted receiver to be compatible with most hands, wrists and elbows that are on the market. Vaskov says this is where much of the engineering is going. “We want to be agnostic to the terminal device,” he said.
Currently, there is a specific code for each manufacturer. “We need to be in constant communication with the prosthetic manufacturers—to make sure our system’s compatible with their [prosthetic devices],” Vaskov said. “Our role in the ecosystem is to provide that interface between the patient and the [device].”
Marrying Tried-and-True with Novel Technology
Using pre-validated components such as the intramuscular electrodes that have been on the market for decades (think pacemaker-style implants), along with the new technology that is used to connect to the nerves and interpret the signals, is what Vaskov says he is confident will give users reliable and long-term performance.
Once the electrodes are implanted into the muscles, RPNI or TMR to read nerves signals that are processed through the sensing unit to the socket-receiver, a wireless charger included with the other components of the system recharge the sensing unit, “similar to how…deep brain stimulators or other implants are charged nowadays,” he said.
The final component is the software, which will be used to calibrate the machine learning (ML) or the control algorithm on the socket receiver. In the beginning, it is used for mapping between signals and movements, which will be different for each person. The careful selection of ML algorithms are essential, as they play a vital role in enabling individuals to control independent and simultaneous movements without false activations, Vaskov said.
The software architecture will include cloud storage for data management and a mobile app for patients and clinicians to interact with the system. The app allows users to monitor control signals, calibrate algorithms and practice controlling virtual hands before actual prostheses, facilitating adaptation and mastery of the system, Vaskov explained.
READ MORE: Robots Open World of Opportunities for A3 President
Continued Innovation for Prosthetic Control Systems
The future of prosthetic control systems looks promising, with advancements focusing on restoring more naturalistic control for users. This includes moving toward intuitive control mechanisms that mimic natural movements rather than preset grips. As control technology evolves, prosthetic limbs themselves are expected to become more advanced, durable, lightweight and energy efficient to meet user demands. With ongoing improvements in control algorithms and prosthetic design, the overall prosthetic care system is set to enhance, offering users greater functionality and usability in their daily lives. “It’s going to be really interesting to see how patients are able to use those devices in their daily lives,” Vaskov said. “I think it’s going to be fascinating.”
On the manufacturing front, 3D printing is playing a significant role in creating components like socket receivers for prosthetic devices. Vaskov said the use of high-quality 3D printing for housings is preferred due to its capabilities and the ability to ensure quality and low volume production. This approach allows for customization and efficient production of components, reducing the need for traditional manufacturing methods like injection molding.
Regarding clinical trials and market timeline, while it is still a developing area, the aim is to have bench-tested systems by 2025 to start clinical trials. The path from trials to market approval involves navigating regulatory processes, making the timeline uncertain, but Vaskov says that progress is underway.
He added that there are possibilities for expanding the technology to benefit patients with congenital limb differences. “It requires further research and testing to better understand and evaluate the benefits of our technology for those patients,” he said. They also are exploring sensory feedback integration through force sensors.
Future iterations may also extend the technology to lower-limb prosthetics, enabling voluntary control of robotic legs using nerve muscle interfaces and customized software, he said: “These efforts are still in the early feasibility and research phase, but are on the horizon.”
READ MORE: Rising Demand for Medical Devices Fueling Market Growth
About the Author
Sharon Spielman
Technical Editor, Machine Design
As Machine Design’s former technical editor, Sharon Spielman produced content for the brand’s focus audience—design and multidisciplinary engineers. Her beat included 3D printing/CAD; mechanical and motion systems, with an emphasis on pneumatics and linear motion; automation; robotics; and CNC machining.
Spielman has more than three decades of experience as a writer and editor for a range of B2B brands, including those that cover machine design; electrical design and manufacturing; interconnection technology; food and beverage manufacturing; process heating and cooling; finishing; and package converting.
Email: [email protected]
LinkedIn: @sharonspielman
Facebook: Machine Design
YouTube: @MachineDesign-EBM
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: