Crystal Structures Key to Steel’s Design Flexibility
This article was updated Feb. 7, 2023. It was originally published Nov. 15, 2002.
RELATED
Advanced High-Strength Steels Add Strength and Ductility to Vehicle Design
Martensitic Stainless Steel Makes Inroads in the Automotive Sector
Understanding the Basics of Amorphous-Iron Motors
Single-Crystal Turbine Blades Earn ASME Milestone Status
Metalworkers and metallurgists have long appreciated the ability to tailor the performance characteristics of steel (an alloy of iron and carbon), including their strength, hardness, ductility and machinability. One way they modify the steel and come up with new types of it is by adding different alloys such as boron, nickel, chromium, manganese, silicon and vanadium. The other way is to alter its crystal structure and makeup by heating, cooling and working it. Both let ferrous alloys be developed to meet a wide variety of design needs.
Here’s a look at how material engineers fine-tune the properties of steel alloys.
In the iron-carbon alloys, an important phase transformation (the A3 transformation) takes place between about 1,300 and 1,600°F. The exact temperature is determined by how much carbon and other alloying elements are in the alloy metal.
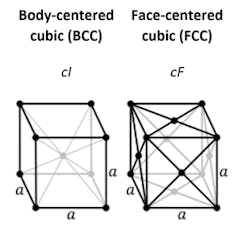
Iron goes from a face-centered cubic (FCC) structure—the gamma phase, or austenite—at high temperatures to a body-centered cubic (BCC) structure—alpha phase, or ferrite—at a lower temperatures. In pure iron, this transformation (the A3 transformation) is marked by a distinct increase in length as the metal cools below the critical temperature because the body-centered lattice is less compact than the face-centered lattice.
High-temperature austenite, an FCC structure, has enough space for carbon atoms to squeeze in between the iron atoms. Iron atoms keep their place on the lattice while carbon atoms become “interstitials.” In low-temperature ferrite, or BCC structure, however, there is no room for carbon atoms. What happens to these carbon atoms determines many of the properties of iron and steel.
For example, while slowly cooling a low-carbon (0.20% carbon) steel such as AISI 1020, transformation starts as the metal reaches 1,555°F. The first metal to reach this temperature turns into ferrite, which expels the interstitial carbon into what’s left of the austenite. As the metal continues cooling, more iron turns into ferrite, leaving less austenite and more regions rich in expelled interstitial carbon.
Finally, at about 1,350°F, the lower end of the transformation temperature range for 1020 steel, the last remaining austenite tries to transform in spite of the rich carbon concentrations. At this point, the carbon bonds with available iron atoms to form Fe3C, an intermetallic compound called cementite (or iron carbide). It precipitates out as a discrete structure and the remaining austenite transforms to ferrite.
The result of this final transformation is a laminate called pearlite. It consists of alternating layers of ferrite and iron carbide. Of course, the portions of metal that transformed previously remain as large islands of pure ferrite. The combined structure of ferrite and pearlite is soft and ductile, but it’s one of the weakest (low strength) form of steel.
In contrast, when ferrous alloys cool rapidly (quenched), expelled carbon atoms have no time to get away from the iron as it turns to ferrite. The steel becomes so rigid that, before the carbon atoms can move, they are trapped in the lattice as the iron atoms try to transform to a BCC structure. The result is a body-centered tetragonal structure with carbon as an interstitial member. Steel that undergoes this type of transformation is martensitic. Naturally, martensite is in a state of non-equilibrium, but its high strength and hardness (and lower ductility) are due to its distorted, stressed lattice structure.
A number of heat-treatment cycles have been developed to alter the structure of steel. For example, when martensite is tempered (heated below A3 temperature) some internal stresses are relieved, and the resulting structure has more ductility than as-quenched martensite.
Other heat treatments change the proportions of pearlite and martensite; some even entrap austenite at room temperature. Others alter or reduce the grain size or their pattern, giving the alloy more strength or toughness.
About the Author
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: