New Technique Helps You Cut Metals Easier
Metallurgy is an interesting science. You would think for the thousands of years humans have been working with it we’d have it pretty well figured out. Well, recently Purdue published a paper titled “Material-Independent Mechanochemical Effect in the Deformation of Highly-Strain-Hardening Metals,” which presented the idea of improving the plastic-flow in the cutting of metal.
Is this some new nanotechnology that takes advantage of Van der Waal’s forces to magically alter the effects of the metal to the designer will? No. Some scientist just drew on different metals with a Sharpie and glue sticks. Oddly enough, though, it did affect the surface energy and cutting forces significantly.
Something as simple as applying a Sharpie to a piece before cutting can make difficult metals easier to process. The paper noted the observations of the physical absorption (adhesion) of the media to the metal, and how that effected the occurrence of an unsteady plastic-flow mode—sinuous flow—characterized by large-amplitude folding and large strains. Essentially, the conclusion was that the better the media absorbed, lower the cutting forces were and flow was improved.
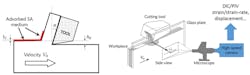
Shown is a schematic of the cutting configuration used in the experiments. The surface-active media is applied to the workpiece free surface remote from the tool-chip interface. The deformation zone is observed in situ to obtain high-resolution quantitative flow-field information.
It is well known that soft metals, as well as metals with a large strain-hardening capacity—such as pure aluminum, iron, copper, and tantalum—are notoriously difficult to cut. This difficulty is shown by large forces, thick chips, and a surface defects. such difficult to cut metals are often called “gummy” metals.
Recently, it was shown that cutting difficulty is due to an unsteady mode of large-strain plastic deformation—sinuous flow, characterized by plastic buckling and large-amplitude material folding—that prevails during cutting. Surface preparation is important in many metal processes, and Purdue decided to revisit surface-active chemical media (media with a strong absorbed layer on metal surfaces and influence surface energies). For this Purdue experimented with marking inks and glue sticks.
The simple act of drawing on the surface of a gummy metal with (for example) a Sharpie helped reduce the difficultly of cutting these materials. Preliminary experiments on pure copper did show a significant reduction in cutting force, but the effects appeared to depend on the strain-hardening of the metal. Annealed copper showed significant effects by adding surface-active media. This paper studied:
- The effects of various chemical media, characterized by different adhesive coupling strengths to metal surfaces, on plastic flow and cutting.
- How these effects are influenced, if at all, by the initial hardening state (e.g., annealed or pre-hardened) of the metal.
|
Materials |
Hardness |
|
Annealed Copper |
68 Vickers |
|
Annealed Iron |
90 HV |
|
Annealed Aluminum |
23 HV |
|
Half-hard (H14) Aluminum |
30 HV |
The surface-active chemical media was classified into three groups:
Group I Shows strong physical adsorption to all three metals.
- Scotch restickable glue stick
- Scotch Super Glue Gel
- Gorilla Super Glue
- Sharpie permanent marker
- Dykem
- Paper Mate Liquid Paper correction fluid
Group II consists of media that show chemical affinity (specificity) for Al only, as established by their ability to corrode oxide-free Al surfaces.
- Isopropyl alcohol (Fisher Chemical)
- Ethanol (Decon Labs)
- 1-butanol (Fisher Chemical).
Group III consists of media that are not known for any significant adsorptive or other similar interactions with any of the metals.
- Distilled water
- Toluene (Consolidated Chemical)
- Acetone (Fisher Chemical)
In addition, there was some other chemical media that didn’t fit into these groups: soap (Proctor & Gamble), paraffin wax (Gulf Wax), adhesive tape (Scotch transparent tape), and diacetone alcohol (Lab Alley). The test was performed under similar cutting parameters and tooling. A high-speed camera was set up to capture the flow during deformation, both with and without surface-active media.
Result
Cutting Force
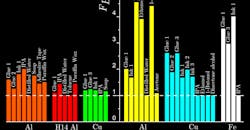
Group-I media show consistent mechanochemical effects and force reductions of 50% independent of the metal to which they were applied. Group-II media show pronounced effects only with some metals. For instance, alcohols in group II—isopropyl alcohol (IPA), butyl alcohol, and ethanol—show the largest force reductions (nearly 80%) with aluminum, yet had no effect with Copper. Isopropyl alcohol, in particular, is the only common ingredient of media in group I (e.g., inks 1 and 2), yet shows no effect with Cu and Fe (unlike inks 1 and 2 themselves).
The results show Group-I media fall around the FC = 0.5 FB line, Group-II media on the FB = 0.2 FC line, and Group-III media around the FB = FC line (negligible mechanochemical effect). Al and Cu refer to metals in the annealed state.
This result is reflective of the strength or weakness of local adhesive bonds that form at the media-metal interface: The alcohols are known to form strong bonds with Al to such an extent that they may even corrode it to form the corresponding alkoxide. Likewise, distilled water and toluene (group III, little-to-no adhesion) show no effects with any of the metals.
Surface Defects and Topography
Alongside the transition in flow mode and the attendant force reduction, the surface-active media application is found to result in a remarkable improvement in quality of the newly created (cut) workpiece surface. As has been demonstrated clearly elsewhere, the occurrence of folding and sinuous flow is synonymous with the formation of pits, cracks, and tears on the resulting surface.
Discussion
Purdue’s experimental results also show that in addition to the strong adsorptive interaction between the media and the metal, a second condition is critical for the mechanochemical effect to occur. This condition is the need for sinuous flow, an unsteady flow mode, to prevail in the deformation zone. This sinuous flow is characterized by large-amplitude material folding besides large strains.
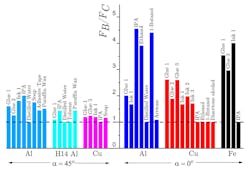
When the sinuous flow is reduced or suppressed by changing the deformation geometry (angle of tooling) to α = 45 deg. (from α = 0 deg.), or by prehardening the workpiece (H14 Al), the extent of the force reduction is significantly smaller or absent altogether. Under these conditions, smooth laminar (steady) flow becomes prevalent. The mechanochemical effect is now much smaller—indeed, in most cases, negligible—pointing to a strong coupling of the media action to the flow type.
The ratio of cutting force for uncoated (FB) to surface-active media coated (FC) workpiece samples for various surface-active media-metal combinations and cutting conditions. Mechanochemical effects result in force reductions of as much as 80% (Al-IPA, α = 0◦). Al, Cu, and Fe refer to metals in the annealed state, while H14 is a half-hard condition for Al.
The mere presence of the surface-active media on the surface induces the metal to behave in a brittle manner during the deformation, when sinuous flow prevails, as with the annealed metals. The reason for this appears to be that sinuous flow produces stress-concentration features, locally, in the deformation zone that trigger local brittle behavior of the metal when the SA media is present
Given that glues and inks adhere to nearly all metals, the beneficial effects of surface-active media demonstrated herein should open up new opportunities in cutting, stamping, piercing, and forming of soft and highly-strain hardening metals such as Fe, Al, Ta, stainless steels, and Ni alloys. Most of these metals are commonly used in everyday life. Finding easier more efficient ways of processing them could reduce a company’s cost and increase the product quality. While this might not be some paradigm shifting nanobots of the future, Purdue demonstrated that sometimes a better mousetrap is already sitting under our noises.
About the Author
Jeff Kerns
Technology Editor
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: