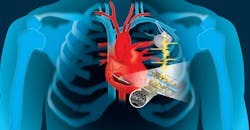
Piezo-Based Energy Harvesting for Medical Implants May Transcend Alternative Approaches
Energy harvesting from the human body (approximately 100 W of consumption at rest) in various forms appears to be a near-perfect power source fit for implanted medical devices, but practical issues have impeded its adoption as a solution. Funded by a five-year NIH Director's Transformative Research Award, a research team at the Thayer School of Engineering at Dartmouth College worked with UT Health San Antonio (part of the University of Texas) and developed a new way to build a piezo-based harvesting transducer for these medical devices.
Their approach uses a combination of thin-film energy-conversion materials with a minimally invasive mechanical design. The work and results are detailed in their paper “Flexible Porous Piezoelectric Cantilever on a Pacemaker Lead for Compact Energy Harvesting” published in Advanced Materials Technologies. Here’s a one-minute video that also demonstrates it:
Providing implanted power is a formidable challenge, noted Dartmouth engineering professor John X.J. Zhang, “How do you create an effective energy source so the device will do its job during the entire life span of the patient, without the need for surgery to replace the battery?” Research associate Lin Dong added, “Of equal importance is that the device not interfere with the body’s function. We knew it had to be biocompatible, lightweight, flexible, and low profile, which also makes it not only fit into the current pacemaker structure, but also scalable for future multifunctionality.”
To build the harvester transducer, the researchers used a combination of thin-film energy-conversion materials with a minimally invasive mechanical approach in a modified pacemaker design. They harnessed the kinetic energy of the lead wire that’s attached to a beating heart, and then converted it into electricity to continually charge the batteries (Fig. 1). The power-generating material, a specialized polymer piezoelectric film called polyvinylidene fluoride-trifluoroethylene (PVDF-TrFE), is designed with porous structures and then built into either an array of small beams or a flexible cantilever.
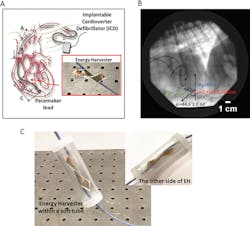
1. Shown is the concept of piezoelectric thin-film energy harvester for an implantable cardioverter defibrillator and a flexible porous PVDF-TrFE dual-cantilever energy harvester on the AICD lead (A). Image from video analysis of a chronically implanted pacemaker lead from a dog (B). A dual-cantilever energy harvester within a soft tube on the pacemaker AICD lead (C). (Source: Thayer School of Engineering at Dartmouth College)
The team created a dual-cantilever structure that wraps around the pacemaker’s lead, with the structure’s two free ends available for connection and subsequent energy collection (Fig. 2). The maximum output was 0.5 V at 43 nA at 1 Hz, a little over 20 nW. By adding a small proof mass of 31.6 mg on the tip of the dual-cantilever tip, the power output increased by a little over 80%—the added mass enabled a larger bending curvature, resulting in higher electrical output from the harvester.
2. Illustrated are a schematic of the porous piezoelectric-energy harvester (A); flexible porous PVDF-TrFE thin film (B); and scanning electron microscope image of the cross-section of the thin film (C). (Source: Thayer School of Engineering at Dartmouth College)
For comparison, today’s ultra-low-power implantable biomedical devices require 0.3 μW for cardiac-activity sensing, 10 to 100 μW for pacemakers, 100 to 2000 μW for cochlear implants, and 1 to 10 mW for neural recording. An advantage of this design is that it’s scalable: two (or more) units can be connected in parallel for a corresponding increase in power output. Initial testing is done using a mechanical shaker, of course, to simulate the motion of the myocardium and the corresponding deformation of a pacemaker lead (Fig. 3).
3. This test setup is based on reported anatomical values of an average adult’s cardiovascular system; a 21-cm long soft tube was used to represent the superior vena cava (SVC) (A). The shaker-based test platform simulates the motion of myocardium (B). (Source: Thayer School of Engineering at Dartmouth College)
The NIH Director's Transformative Research Award is part of the High-Risk, High-Reward Research program supporting “individuals or teams proposing transformative projects that are inherently risky and untested but have the potential to create or overturn fundamental paradigms and may require very large budgets.”
About the Author
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: