“DNA Barcoded” Nanoparticles Identify Drug-Delivery Candidates
A new screening process developed at the Georgia Institute of technology could dramatically accelerate the process of identifying nanoparticles suitable for delivering therapeutic RNA into living cells. The technique would let researchers screen hundreds of nanoparticles at a time, identifying the organs in which they accumulate, and verifying they can successfully deliver an RNA cargo into living cells.
Based on work known as “DNA barcoding,” technicians first insert unique snippets of DNA into as many as 150 different nanoparticles for simultaneous testing. The nanoparticles are then injected into animal models and allowed to travel to organs such as the liver, spleen, or lungs. Next, genetic sequencing techniques identify which DNA-labeled nanoparticles have reached specific organs.
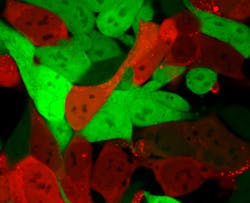
In the upgraded version of the new technique known as FIND (fast indication of nanoparticles discovery), researchers insert a snippet of mRNA that has turned into a protein known as “Cre,” along with the DNA. The Cre protein generates a red glow, identifying cells the nanoparticles have entered and successfully delivered the mRNA drug. This lets researchers easily identify which nanoparticles can deliver RNA drugs to the cells of the specific organs.
Cells that are normally bright green turn bright red after lipid nanoparticles have delivered an mRNA cargo encoding Cre. Red contain the mRNA, while green cells do not. (Credit: Daryll Vanover, Georgia Tech)
The FIND technique would replace in vitro screening, which has limited success at identifying nanoparticle carriers for genetic therapies.
Therapies based on RNA and DNA could address a broad range of genetically based diseases, including atherosclerosis, where such therapies may reverse the buildup of plaque in arteries. Nanoparticles used to deliver RNA and DNA into cells are made from several ingredients whose levels can be varied, creating the potential for tens of thousands of different nanoparticles. Finding the right combination of these ingredients to target specific cells has required extensive trial-and-error discovery processes that have limited the use of RNA and DNA therapies.
Using this DNA barcoding process lets hundreds of possible nanoparticle combinations be tested simultaneously in a single animal. Until now, however, researchers could only tell that the combination had reached specific organs. By examining which cells within the organs have the red glow, they can now verify that the nanoparticles carried the barcodes and delivered functional mRNA drugs into the cells.
Technical challenges ahead include demonstrating that identifying an affinity for mouse organs predicts which particles will work in the human body, and that the approach works for different classes of genetic therapies.
Experimentally, the researchers produce the nanoparticles, which takes about 90 seconds to turn out 250 or so samples. They are examined for proper size range—40 to 80 nanometers in diameter—before being purified and sterilized for injection into the animals.
After three days, the researchers separate cells that are glowing red and sequence the DNA snippets in them to identify which chemical compositions were most successful at entering cells of specific organs. The most promising chemical compositions are used to develop new batches of candidate nanoparticles for a new round of screening, which takes about a week to complete.