MOF Filter Pulls Purified Ethylene from Oil Refinery Soup
An innovative filter made of a metal organic framework (MOF) may soon reduce the environmental cost of manufacturing plastics. It was recently built by engineers from the National Institute of Standards and Technology (NIST) and the University of Texas at San Antonio. The new material efficiently extracts ethylene, the key ingredient in the most common form of plastic, from a mixture of other chemicals.
MOFs are substances that can separate individual hydrocarbons from the soup of organic molecules produced by oil-refining processes. They hold potential value for the plastic and petroleum industries because they could perform complex separations far more cheaply than standard oil-refining techniques.
This promise has made MOFs the subject of intense study at NIST and elsewhere, leading to MOFs that separate different octanes of gasoline or speed up complex chemical reactions. One major goal has proved elusive, though: an industrially preferred method for isolating ethylene, the molecule needed to create polyethylene, the plastic used to make shopping bags and other everyday containers.
Credit: W. Zhou/NIST
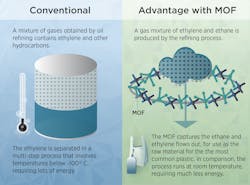
Making plastic takes lots of energy. Ethylene, one of the many hydrocarbon molecules found in crude oil refining, must be highly purified for the manufacturing process to work. But the current method of separating ethylene from other hydrocarbons is a high-energy process that cools crude to lower than −100°C.
Ethylene and ethane constitute the bulk of the hydrocarbons in the mix, and separating these two is by far the most energy-intensive step. Finding an alternative way to separate them would reduce the energy needed to make the 170 million tons of ethylene manufactured worldwide annually.
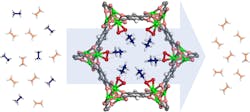
Scientists have been searching for such an alternative method for years, and MOFs appear promising. On a microscopic level, they look a bit like half-built skyscrapers of girders with no walls. The girders have surfaces covered by certain hydrocarbon molecules which stick to firmly to them. Pouring a mix of two hydrocarbons through the right MOF pulls one kind of molecule out of the mix, letting the other hydrocarbon emerge in pure form. The trick is to create a MOF that lets ethylene pass through. For the plastics industry, this has been the sticking point.
“It’s difficult to do,” says Wei Zhou, a NIST researcher. “Most MOFs that have been studied grab onto ethylene rather than ethane. A few even demonstrate excellent separation performance and selectively adsorb ethylene. But from an industrial perspective, we prefer to do the opposite if feasible. We want to adsorb the ethane byproduct and let the ethylene pass through.”
The research team spent years trying to crack the problem. In 2012, another NIST research team found that a particular framework dubbed MOF-74 was good at separating a variety of hydrocarbons, including ethylene. It seemed like a good starting point, and the team members scoured the scientific literature for additional inspiration. An idea taken from biochemistry finally sent them in the right direction.
“A huge challenge in chemistry is finding ways to break the strong bonds that form between carbon and hydrogen,” says Banglin Chen, a University of Texas at San Antonio professor who led the team. “Doing that lets you create a lot of valuable new materials. We found previous research that showed compounds containing iron peroxide could break that bond.”
The team reasoned that to break the bond in a hydrocarbon molecule, the compound would have to attract the molecule in the first place. When they modified MOF-74’s walls to contain a structure similar to iron peroxide, it attracted ethane.
The team used neutron diffraction to explore MOF-74’s atomic structure and determine what part of it attracts ethane. Although the team’s modified MOF works efficiently, it may require some additional development before seeing action in a refinery.