Researchers Add (and Take Away) Ions from a Quantum Crystal
Researchers at Purdue University have shown how to shuttle lithium ions back and forth into the crystal structure of samarium nickelate, a quantum material, opening the door to a representing a new avenue for research and potential applications in batteries and brain-inspired computers containing artificial synapses. Samarium nickelate crystal is in a class of quantum materials called strongly correlated electron systems, which have exotic electronic and magnetic properties, and has characteristics that tap into quantum mechanical interactions.
Adding lithium ions make the crystal expand and increases its conduction of the ions. The effect also works with other types of ions, particularly sodium ions, so it might have applications in energy storage.
“The results highlight the potential of quantum materials and in the design of ion conductors,” says Shriram Ramanathan, a Purdue University professor of materials engineering who is leading the research. “There is a lot of research to identify solid-state ion conductors for building batteries, for example.
“We showed that this general family of materials can hold these ions and established some general principles for designing these sorts of solid-state ion conductors,” Ramanathan explains. “We showed that ions such as lithium and sodium can move through this solid material, and this opens up new directions for research.”
To add the ions, researchers applied a voltage which let the ions occupy spaces between atoms in the material’s crystal lattice. Moreover, the ions remained in place after the current was turned off, a “non-volatile” behavior that might be harnessed for computer memory. This could be a more efficient way to store and conduct electricity.
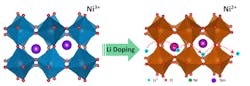
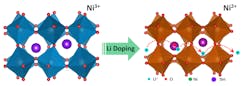
This graphic shows lithium ions inserted into the crystal structure of a quantum material called samarium nickelate, suggesting a new avenue for research and potential applications in batteries, “smart windows” and brain-inspired computers containing artificial synapses. (Purdue University image/Yifei Sun)
Adding lithium ions to the crystal structure also changes the material’s optical properties, suggesting potential applications such as coatings for “smart windows” whose light transmission properties are altered when voltage is applied.
The research findings demonstrated behavior related to the “Mott transition,” a quantum mechanical effect describing how adding electrons changes the conducting behavior of some materials. “As we add more electrons to the material, it becomes less and less conducting, which makes it a very interesting system to study, and this effect can only be explained through quantum mechanics,” Ramanathan says.
Calculations showed that undoped samarium nickelate is a narrow-gapped semiconductor, meaning it is not metallic, but electrons can still be excited into a conducting state without much effort. As lithium is added to samarium nickelate, the lithium ions bind to an oxygen and an electron localizes on a nearby nickel-oxygen octahedron. When an electron has localized on every nickel-oxygen octahedron, the material changes into an insulator. This is a rather counterintuitive result: Adding electrons makes the material more insulating.