Why High-Energy Batteries Fail and Start Fires
Scientists from Stanford University and the Department of Energy’s SLAC National Accelerator Laboratory have captured the first atomic-level images of finger-like growths (dendrites) that pierce the barrier between battery compartments, triggering short circuits or fires. Dendrites and the problems they cause have been a stumbling block on the road to developing new types of batteries that store more energy so electric cars, cell phones, laptops, and other devices can go longer between charges.
The researchers imaged the inner lives of batteries for the first time with cryo-electron microscopy, or cryo-EM, a technique that can image delicate, flash-frozen proteins and other “biological machines” in atomic detail.
The new images reveal that each lithium metal dendrite is a long, well-formed six-sided crystal, not the irregular pitted shape depicted in previous electron microscope shots. The ability to see this level of detail for the first time gives researchers a powerful tool for understanding how batteries and their components work at the most fundamental level, as well as for investigating why high-energy batteries sometimes fail.
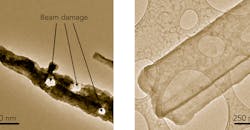
This room-temperature TEM image (left) shows how exposure to air has corroded a lithium metal dendrite and the electron beam has melted holes in it. In contrast, a cryo-EM image of a dendrite (right) shows that freezing has preserved its original state, revealing that it’s a crystalline nanowire with well-defined facets.
The team managed to get video of a dendrite curling up and melting when hit with an electron beam during transmission electron microscopy (TEM), which takes place at room temperature. It was also able to zoom in on a flash-frozen dendrite magnified about 400,000 times during cryo-EM imaging. Freezing protects it from the electron beam so researchers could see its intact structure for the first time, down to an atomic level.
“This is super exciting and opens up amazing opportunities,” says Yi Cui, a professor at SLAC and Stanford and investigator with the Stanford Institute for Materials and Energy Sciences (SIMES) whose group did the research.
“With cryo-EM, you can look at a material that’s fragile and chemically unstable and you can preserve its pristine state, what it looks like in a real battery, and look at it under high resolution.
“This includes all kinds of battery materials,” he adds. “The lithium metal we studied is just one example, but it’s an exciting and very challenging one.”Cui’s lab is developing strategies to prevent damage from dendrites, such as adding chemicals to the electrolyte to keep dendrites from growing, or developing a “smart” battery that automatically shuts off when it senses dendrites invading the barrier between the battery’s chambers. But until now, scientists could not get atomic-scale images of dendrites or other sensitive battery parts. The method of choice, TEM, was too harsh for many materials, including lithium metal.
“TEM sample preparation is carried out in air, but lithium metal corrodes very quickly in air,” notes Yuzhang Li, a Stanford grad student who led the work with fellow grad student Yanbin Li. “Every time we tried to view lithium metal at high magnification with an electron microscope, the electrons would drill holes in the dendrite or even melt it altogether.
With cryo-EM, researchers could see the way electrons bounced off the atoms in the dendrite, revealing the locations of individual atoms (left). They were even able to measure the distance between atoms (right). The spacing between these atoms shows that they are lithium.
“It’s like focusing sunlight onto a leaf with a magnifying glass,” Yuzhang Li continues. “But if you cool the leaf at the same time you focus the light on it, the heat will be dissipated and the leaf will be unharmed. That’s what we do with cryo-EM.”
In cryo-EM, samples are flash-frozen by dipping them into liquid nitrogen, then sliced for examination under the microscope. This lets researchers freeze a whole coin-cell battery at a particular point in its charge-discharge cycle, remove the component they’re interested in, and see what is happening inside that component at an atom-by-atom scale. They could even create a stop-action movie of battery activity by stringing together images made at different points in the cycle.
For this study, the team used a cryo-EM instrument at Stanford School of Medicine to examine thousands of lithium metal dendrites that had been exposed to various electrolytes. It looked not only at the metal part of the dendrite, but also at a coating called SEI, or solid electrolyte interphase, that develops as the dendrite reacts with the surrounding electrolyte. This same coating also forms on metal electrodes as a battery charges and discharges, and controlling its growth and stability are crucial for efficient battery operation.
The researchers discovered, to their surprise, that the dendrites are crystalline, faceted nanowires that prefer to grow in certain directions. Some of them developed kinks as they grew, but their crystal structure remained surprisingly intact in spite of the kinks.
Zooming in, they used a different technique to look at the way electrons bounced off the atoms in the dendrite, revealing the locations of individual atoms in both the crystal and its SEI coating. When they added a chemical commonly used to improve battery performance, the atomic structure of the SEI coating became more orderly. The team says this may help explain why the additive works.
“We were really excited,” says Yanbin Li. “This was the first time we were able to get such detailed images of a dendrite, and we also saw the nanostructure of the SEI layer for the first time. This tool can help us understand what different electrolytes do and why certain ones work better than others.”
Going forward, the researchers intend to learn more about the SEI layer’s chemistry and structure.