A Primer on Valves Used in Chromatography and Spectrometry Analysis
READ MORE: Medium-Pressure Ball Valves Provide a Rugged Solution
Equipment for gas chromatography, liquid chromatography and mass spectrometry is complex, with multiple subsystems that control the flow of sample analyte, carrier gas and solvent fluids. The performance and accuracy of these devices depend on the precision and repeatability of various types of valves that must handle different gases and liquids at a range of flow rates. Leak-free operation and cleanliness are also critical considerations.
The following is a review of the valves used in the three types of analysis systems along with an explanation of their role in ensuring the highest levels of performance and productivity.
Valve Basics
Analytical instruments typically use valves with different technology; these four are the common: isolation, pinch, proportional and general-service valves.
Isolation valves prevent contamination of various liquids and/or gases in a system. A sealing mechanism keeps the fluid separate from the actuator and prevents it from coming into contact with the valve control surfaces, ensuring that the sensitive media within the valve body remains pure.
A variety of sealing techniques may be used, ranging from a simple diaphragm to more sophisticated flapper, bellows, lever or rocker seal designs. These sealing options make the valves versatile enough to handle and isolate virtually any type of media while protecting it from contamination. In addition, these valves are designed to control and deliver very low internal fluid volumes and to be easy to clean for reuse. These technical advantages make isolation valves a primary choice for fluid handling in equipment used for research and analytical purposes.
Pinch valves represent a specific type of isolation valve used to control the flow of a media that is carried in a flexible tube that passes through the valve body. The valve actuator and body surround and compress, or “pinch,” the flexible tube to control media flow. When open, pinch valves ensure a free flow throughout the tubing. Because of their hermetic separation and flow characteristics, pinch valves are ideal for handling all kinds of sensitive media, with the benefit of media non-contamination and fast tubing change-out.
READ MORE: Piezo-Electric Valves for Hydraulic and Pneumatic Systems
Proportional valves are solenoid-driven valves that respond precisely to the slightest changes in electrical input signals, producing small, proportional movements of the actuator and making fine adjustments to the flow path for extremely accurate regulation of both gas flow and pressure. Compact proportional valves are ultra-reliable, delivering speed and precision. Because the actuator is designed to minimize friction and the valve body is able to handle varying input pressures, proportional valves are capable of regulating a wide range of flow rates, including the low flow rates (just a few milliliters per minute) required in gas-chromatography or mass spectrometry applications.
General-service valves typically do not offer the design characteristics for physical or hermetic separation of valve actuators from fluids they control. However, they can be built with biocompatible materials (e.g., O-rings, seals, valve bodies) that meet material compliance requirements for “wetted” surfaces. Therefore, they are quite versatile and are known for excellent stability and long cycle life under continuous duty.
So, how are these valves used in gas and liquid chromatography and mass spectrometry?
Gas and Liquid Chromatography
These two analytical techniques serve the same purpose—i.e., separating the components of any mixture. Gas chromatography separates and analyzes the compounds in their volatile or gaseous phase. In contrast, the liquid chromatography technique separates the dissolved ions, particles or molecules in a liquid.
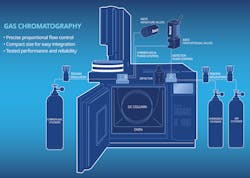
In gas chromatography, an inert gas is used to carry samples. Helium, nitrogen and hydrogen are commonly used as the carrier gas. The sample to be analyzed is injected into the injection port and carried into the column. The sample then is separated inside the column and different chemical compounds are detected by a specific type of detector. Computerized or electronic pneumatic control (EPC) systems are widely used in modern gas chromatographs (GC). There are two main purposes for EPC manifold: One is to control carrier gas flow, and the other is to control detector gas (air and hydrogen) for flame ionization detection (FID).
In both functions, solenoid-driven proportional valves make fine adjustments of the flow path for extremely accurate gas flow control and are critical to ensure chromatography analysis reliability and repeatability. These valves with small orifice sizes readily handle the low flow rates (just a few milliliters per minute) required in most gas chromatography applications. Miniature general-service valves with compact size and high tightness will also be used to on/off control gas sampling, purge flow or vent the system.
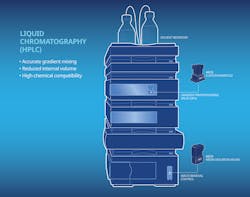
Liquid chromatography also involves two distinct flow control operations. Upstream, the low-pressure-mixing system will use proportioning valve to blend the solvents. Various proportioning valves open and close to allow the right proportion of each solvent (up to four) to enter the mixer. Specific combinations of solvents (called gradients) are used depending on the sample being analyzed. The proportioning accuracy with which each solvent is dispensed is critical because it affects the ultimate precision of the analysis and can reduce cycle time. Compact size, low internal volume and extremely short response time are other important features.
After the sample compound is injected into the solvent stream, it is pumped to the chromatography column, where it will be separated into individual components. The constituent elements are then recognized and analyzed by a detection unit. Two- or three-way isolation valves are commonly used to control the wash solutions and waste drainage for system cleaning and waste disposal safety.
Mass Spectrometry
Mass spectrometry is another powerful analytical technique that measures the mass-to-charge ratio (m/z) of charged particles. It is used for determining masses of particles and for determining the elemental composition of a sample or molecule. The analysis results are presented as a mass spectrum, or a plot of intensity as a function of the mass-to-charge ratio.
READ MORE: How to Choose the Right Proportional Pressure Control Valves
When a sample is introduced into a mass spectrometer, it is first evaporated at high temperatures, and then ionized to become either positively or negatively charged. Nitrogen gas is used to aid in solvent transportation and sample nebulization and evaporation. On/off or proportional valves provide proper flow control of the nitrogen gas, ensuring the stability and sensitivity of the mass spectrometry results. A low flow rate (around 0.2 liters per minute) is required for maximum accuracy.
Next, a “collision gas” such as argon is introduced into the collision cell where ions are broken into fragments as they collide with the argon molecules. Proportional valves control the argon supply into the collision cell. All mass spectrometers operate at very low pressure (high vacuum). Therefore, the valve’s ability to achieve and maintain an effective vacuum is vital to the process. General-service valves with high tightness and vacuum operation capability allow the vacuum manifold to be vented and refilled with dry nitrogen or filtered air.
Top of Mind
In this article, words like “precise,” “accurate,” “reliable” and “repeatable” have been used frequently due to their importance in chromatography and spectrometry operations. These operations must perform at submolecular levels with acuity, so valve design and materials used in construction must be evaluated carefully. Valves must also be robust, ensuring a long working lifetime without deteriorating in performance or requiring frequent maintenance or replacement. Designers should keep these factors top of mind when choosing valves for gas and liquid chromatography and mass spectrometry equipment.
Silvia Shen is the global product marketing manager for industrial analyzers for Emerson’s Analytical and Medical Segment. She has more than five years of experience in the area of analytical instruments.
About the Author
Silvia Shen
Global Product Marketing Manager, Industrial Analyzers, Analytical & Medical, Emerson Automation Solutions
Silvia Shen is the Global Product marketing manager, Industrial Analyzers, Analytical & Medical for Emerson Automation Solutions. She has more than five years’ experience in the area of analytical instruments.
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: