Choosing the Right Battery to Power a Remote Wireless Device
This file type includes high resolution graphics and schematics when applicable.
The world is becoming increasingly wireless as evidenced by the rapid emergence of the Internet of Things (IoT) and the development of low-power communications protocols such as ZigBee, Bluetooth, DASH7, INSTEON, and Z-Wave. These energy-saving protocols, in conjunction with energy-saving chip design, permit certain wireless devices to operate maintenance-free for up to 40 years.
Consumer wireless devices have proliferated and the market for industrial wireless devices has expanded just as rapidly. Wireless technology is being applied to a wide range of applications, including automated meter reading (AMR) and automated metering infrastructure (AMI), wireless mesh networks, structural sensors, machine-to-machine (M2M) and system control and data acquisition (SCADA), data loggers, measurement while drilling, oceanographic measurements, and emergency/safety equipment, to name a few. In terms of choosing the right power supply, industrial-grade devices typically involve far more complex technical challenges than consumer grade electronics, including:
- Reliability: Remote sensors can be located in hard-to-reach areas, making battery replacement difficult. Bad batteries should not interrupt the data communications from the sensor.
- Long operating life: The self-discharge rate of the battery can be more than the device usage of the battery. Initial battery capacity should be as high as possible.
- Wide operating temperatures: Industrial devices tend to be in extreme cold or hot places.
- Small size: Battery-powered instruments have to be able to be fit anywhere. Battery energy density should be high.
- Voltage: The higher the voltage required, the lower amount of cells needed.
- Lifetime costs: Replacement costs over time must be taken into account.
Identifying the Ideal Power Supply
A remote wireless device is only as reliable as its power supply, the needs of which are optimized based on application-specific requirements. While primary (non-rechargeable) batteries power the vast majority of remote wireless devices, certain applications may be ideally suited for energy-harvesting devices used in conjunction with rechargeable batteries or supercapacitors, which store the harvested energy and deliver the high pulses needed to power advanced communications. The battery-selection process often encompasses the following parameters:
- energy consumed in active mode (including the size, duration, and frequency of pulses);
- energy consumed in dormant mode (the base current);
- storage time (as normal self-discharge during storage diminishes capacity);
- thermal environments (including storage and in-field operation);
- equipment cut-off voltage (as battery capacity is exhausted, or in extreme temperatures, voltage can drop to a point too low for the sensor to operate);
- battery self-discharge rate (which can be higher than the current draw from average sensor use); and
- cost considerations.
Choosing Among Primary Batteries
If long battery operating life is not a major concern, then an alkaline battery may suffice. This is mainly true for consumer products such as flashlights, television remote controllers, and toys where batteries are easily accessible. Alkaline batteries have certain drawbacks, including low voltage (1.5 V or lower), a limited temperature range (0°C to 60°C), a high self-discharge rate that reduces life expectancy to two to three years, and crimped seals that may leak.
The low initial cost of a consumer alkaline battery can sometimes be misleading, as this investment is relatively short-lived and carries downstream risks associated with loss of productivity and/or data due to premature battery failure. Alkaline batteries must be replaced every few months, which raises the total cost of ownership. This is especially true if the wireless device is deployed in a remote, inaccessible location, where the labor costs associated with battery replacement can be substantially higher than cost of the battery.
Lithium remains the preferred choice for remote wireless applications due its intrinsic negative potential, which exceeds that of all other metals. Lithium is the lightest non-gaseous metal, and offers the highest specific energy (energy per unit weight) and energy density (energy per unit volume) of all available battery chemistries. Lithium cells, all of which use a non-aqueous electrolyte, have a normal operating current voltage (OCV) ranging between 2.7 and 3.6 V. The absence of water allows lithium batteries to endure more extreme temperatures.
Numerous primary lithium chemistries are available (see Table 1), including iron disulfate (LiFeS2), lithium manganese dioxide (LiMNO2), lithium thionyl chloride (LiSOCl2), and lithium metal oxide chemistry.
Consumer-grade lithium iron disulfate (LiFeS2) cells are relatively inexpensive, delivering the high pulses required to power a camera flash for example. These batteries have limitations, including a narrow temperature range of -20°C to 60°C, a high annual self-discharge rate, and crimped seals that may leak.
Lithium manganese dioxide (LiMNO2) cells, including the popular CR123A, provide a space-saving solution for cameras and toys, as one 3-V LiMNO2 cell can replace two 1.5-V alkaline cells. LiMNO2 batteries can deliver moderate pulses, but suffer from low initial voltage, a narrow temperature range, a high self-discharge rate, and crimped seals.
Lithium thionyl chloride (LiSOCl2) cells are the preferred choice for remote wireless applications that demand exceptionally long battery life even under extreme environmental conditions. Prime examples include windshield mounted electronic toll tags that must endure the extreme temperature cycling, and environmental sensors deployed in Arctic climates.
Bobbin-type LiSOCl2 chemistry offers the highest capacity and highest energy density of any lithium cell, along with an extremely low annual self-discharge rate; less than 1% per year. Bobbin-type LiSOCl2 batteries feature a glass-to-metal hermetic seal, and deliver the widest possible temperature range (-80°C to 125°C). This extended temperature range is especially critical to applications such as the medical cold chain, where wireless sensors are used to monitor the transport of frozen pharmaceuticals, tissue samples, and transplant organs at carefully controlled temperatures as low as -80°C. Certain bobbin-type LiSOCl2 demonstrate successful operations under prolonged test conditions at -100°C, which far exceeds the maximum temperature range of alkaline cells and consumer grade lithium batteries.
How a bobbin-type LiSOCl2 battery is manufactured, and the quality of raw materials, can greatly affect product performance. For example, certain brands of LiSOCL2 batteries can deliver as little as 10-year operating life with an annual self-discharge rate of 2% to 3% per year, while a different brand using the same chemistry can achieve up to 40-year operating life with a significantly low self-discharge rate of just 0.7% per year. Specifying a battery with the lowest possible self-discharge rate can serve to reduce your total cost of ownership by eliminating the need for future battery replacements.
If a water utility deploying automated meter infrastructure (AMI) was to use longer-life batteries to power thousands of meter transmitter units (MTUs), resulting in the elimination of just one system-wide battery change-out over a 20-year period, it could potentially save millions of dollars without compromising system reliability. Extended battery operating life is especially beneficial for inaccessibly located devices, such as structural sensors attached beneath bridge trusses, as the labor expense and safety rigging required to replace these batteries would far exceed the cost of the batteries themselves.
To achieve 40-year operating life, a bobbin-type LiSOCl2 battery must be manufactured to extremely high quality standards using high grade raw materials, with strict adherence to total quality management tools such as six sigma methodologies and statistical process controls (SPC) to ensure greater lot-to-consistency. Therefore, it is essential to verify the accuracy of all battery manufacturer claims involving battery life expectancy.
Specialized Batteries Deliver High Rate Current
Applications that require continuous high rate power and/or high pulses for a limited amount of time (i.e., surgical power tools, automatic external defibrillators (AEDs), emergency beacons, and smart munitions) demand a unique power management solution, which is now available through the development of lithium metal oxide batteries.
Constructed with a carbon-based anode, a multi-metal oxide cathode, and an organic electrolyte, lithium metal oxide batteries can deliver up to a 20-year operating life with an annual self-discharge rate of less than 1% per year. These small but powerful cells feature a nominal voltage of 4 V and up to 2 Wh of energy, with a discharge capacity of 135 mAh to 500 mAh, capable of handling 5 A continuous loads and 15A maximum pulses. Lithium metal oxide batteries also feature a wide temperature range (-40°C to 85°C), and a hermetic seal.
High-Pulse Applications Offer Unique Challenges
A growing number of wireless devices require high pulses of energy to power advanced two-way communications or remote shut-off capabilities. To conserve energy, these devices remain mainly in a “standby” mode that requires little or no current, periodically switching to an “active” mode that draws medium to high pulses to initiate data acquisition and transmission.
Due to their low rate design, standard bobbin-type LiSOCl2 batteries may experience a temporary drop in voltage when first subjected to this type of pulsed load: a phenomenon known as transient minimum voltage (TMV). One possible way to minimize TMV is to use supercapacitors in conjunction with lithium batteries. However, supercapacitors have major drawbacks for industrial-grade applications, including relatively high self-discharge rates that can cause premature battery failure, and a limited temperature range that prohibits their use in extreme environments. Solutions involving multiple supercapacitors may also require the use of balancing circuits that also need power.
A cost-effective alternative is to combine a standard bobbin-type LiSOCl2 cell with a patented Hybrid Layer Capacitor (HLC). The battery and HLC work in parallel, with the battery supplying long-term low-current power in the 3.6 to 3.9 V nominal range, while the single-unit HLC delivers high pulses, thus avoiding the balancing and current leakage problems associated with supercapacitors. These hybrid batteries also feature a unique end-of-life performance curve; allowing programmed devices to deliver low battery status alerts.
Certain bobbin-type LiSOCl2 modified batteries deliver moderate to high pulses without the use of an HLC, thus delivering high capacity and high energy density without experiencing voltage drop or power delay. These specially modified batteries utilize available capacity efficiently extending battery life up to 15% in extremely hot or cold temperatures.
Considering Harvesting Energy
A growing number of wireless devices are being powered by energy harvested from external sources such as solar, wind, thermal, and kinetic energy, and RF/EM signals (see "Energy Harvesting Supports the Industrial IoT"). The decision to use an energy-harvesting device hinges on numerous factors: the reliability of the device and its energy source; the expected operating life of the device; environmental requirements; size and weight considerations; and total cost of ownership. The amount of harvested energy is often relatively small, especially for devices that draw only a few microamps of current daily.
An energy-harvesting device generally contains five basic components: a sensor, a transducer, an energy processor, a microcontroller, and an optional radio link. The sensor detects and measures environmental parameters such as motion, proximity, temperature, humidity, pressure, light, strain vibration, and pH. The transducer and energy processor work in tandem to convert, collect, and store the electrical energy in either a rechargeable lithium battery or a supercapacitor. The microcontroller collects and processes the data. The radio link communicates with a host receiver or data collection point.
Rechargeable battery technology has evolved, with lithium-ion (Li-ion) and lithium polymer batteries gaining popularity. Lithium polymer batteries are manufactured in thin, flexible sheets, which is ideal for consumer handheld devices that need to be thin and stylish. Rechargeable lithium-ion (Li-ion) batteries are more commonly utilized in industrial applications.
Consumer-grade Li-ion cells are reasonably inexpensive and widely available, but drawbacks include a life expectancy of less than five years and 500 recharge cycles, a moderate temperature range of -10°C to 60°C, and an inability to deliver high pulses.
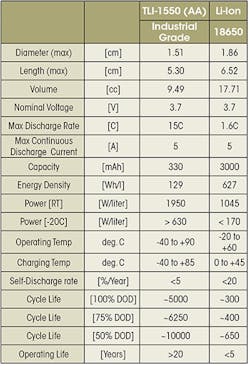
Industrial-grade Li-ion batteries are the preferred choice for long-term deployment in remote and inaccessible locations. They are able to operate for up to 20 years and 5,000 full recharge cycles, provide a much wider temperature range (-40°C to 85°C), deliver high pulses (5 A for an AA-size cell), and feature glass-to-metal hermetic seals, whereas consumer rechargeable batteries use crimped seals more prone to leakage.
Every application is unique, and specific requirements dictate the choice of power supply. With explosive growth in wireless technology, there are abundant opportunities to design autonomously powered devices using advanced primary and rechargeable lithium battery technologies. If the application demands long-term reliability, then the use of an industrial grade battery reduce the long-term cost of ownership.
Sol Jacobs is Vice President and General Manager of Tadiran Batteries.