New Nanosphere Material May Overcome Limits of Lithium Ions
A research team at Rensselaer Polytechnic Institute, Troy, N.Y., has developed a new material—cobalt-oxide meoporous nanospheres—that could overcome two of the major limitations of lithium-ion batteries.
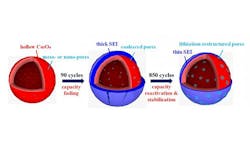
One limitation is mechanical. As the battery charges, lithium ions diffuse into the anode and cause it to expand. Then, during discharges, the ions leave the anode and it shrinks back to its original size. Stresses from the recurring size changes damage the battery and reduce the amount of energy it can hold.
The other limitation, a chemical one, is that the electrolyte reacts with electrons and lithium ions to form a solid-electrolyte interface (SEI) on the electrode. This layer slows the movement of lithium ions in the battery. As the anode grows and shrinks over time, the SEI cracks, then re-forms, and usually gets thicker over time. The thicker SEI hurts battery performance and life.
The cobalt-oxide nanospheres have pores on their surface and are hollow. When an anode made of them is first used, the SEI gets thick and the pores get larger as they grow and shrink. But soon, the SEI starts to get thinner, thanks to the large surface area of the many nanospheres. This makes it easy for lithium ions to pass through. The SEI’s thinness also makes it flexible, so it does not crack and re-form when the nanospheres expand and shrink. Tests show that with anodes of this new material, batteries can withstand thousands of charge-discharge cycles and retain their capacity and life.
If the material can be commercialized, it could lead to lithium-ion batteries being used in high-power applications such as electric cars.