Water Molecules Take on Hexagonal Structure in Nanoscale Spaces
In a paper published in Physics Review Letters, Oak Ridge National Laboratories presents findings for a quantum state of water in which a molecule’s hydrogen atoms occupy six ground states around oxygen at once. What resulted was that their classically bent linear shape transforms to a hexagonal, double-top structure with virtually a zero dipole moment.
The phase is an effect of hydrogen quantum tunneling—caused by tight confinement of individual water molecules in spaces that rival their size. In this experiment, water molecules are confined to the lattice spaces in a beryl gemstone that measure 5 angstroms—about the width of 5 atoms. The team used high- and low-energy neutron scattering to determine the position of hydrogen atoms. Experiments were carried out by the Spallation Neutron Source at the ORNL and at the neutron facility at Rutherford Appleton Laboratory in the U.K.
The report includes computational modeling of this new quantum state, which will be valuable for researching the behavior of water in tight spaces, from nanoscopic crevices in rocks to carbon nanotubules and other man-made nanostructures. It may lead to new discoveries in drug delivery to individual cells using nanoneedles. Based on the neutral polarity of these new molecules, water is bound to act much differently with its surroundings than it does in classical systems.
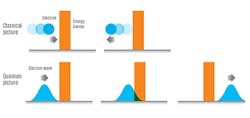
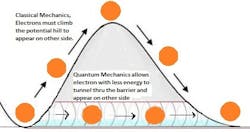
Quantum Tunneling
Also, unlike matter, wave particles can propagate through barriers that would be energetically impermeable to matter. So while a quantum-wave particle may exist in several positions, it exists in a single position when observed as a particle of matter. In turn, observation of the particle on the other side of an energy barrier becomes a probability.
Quantum tunneling, which is a cause of many phenomena, is widely taken advantage of in technology. For example, as transistors continue to exponentially shrink and their barriers get thinner according to Moore’s law, electrons will increasingly tend to leak between the barriers of an off-state transistor. Scientists may try to minimize this effect, or exploit it by altering the thickness of energy barriers for controlled QT.
Moreover, scanning tunneling microscopes leverage QT by introducing a voltage bias that causes electrons to tunnel from subject atoms to the metal scanning tip. This generates a current that can be detected for atomic imaging.
That said, QT can result in a broad range of unexpected molecular configurations, even those that are not classically and energetically favorable. The hydrogen atoms of water trapped in beryl crystal channels spread out or “delocalize” from the central oxygen, shrinking the energy barrier between each configuration down to about 50 meV. In turn, the two hydrogens can more easily tunnel through six separate ground states. Since the probability of each space occupation is the same, the molecule takes the form of a symmetrical hexagon, with each space being occupied at the same time.
The team used low-energy neutrons to detect hydrogen’s position as it tunnels through six separate ground states. They brought the system down to 5 Kelvin to eliminate most thermal effects that could obscure the results of QT. The scattering test identified seven peaks in the scattering spectrum. A second set of experiments used high-energy neutron scattering at Rutherford Appleton Laboratory, revealing kinetic energy that is 30% lower than usual in water’s hydrogen. This proved the “delocalization” theory of hydrogen from oxygen when confined in beryl.
The results from ORNL are consistent with the results of another experiment conducted by Boris Gorshunov of the Moscow Institute of Physics and Technology. In his experiment, terahertz spectroscopy was used to detect the ground energy levels of water in beryl.
About the Author
Leah Scully
Associate Content Producer
Leah Scully is a graduate of The College of New Jersey. She has a BS degree in Biomedical Engineering with a mechanical specialization. Leah is responsible for Machine Design’s news items that cover industry trends, research, and applied science and engineering, along with product galleries. Visit her on Facebook, or view her profile on LinkedIn.
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: