How Does Radiated EMI Impact Medical Devices?
What You'll Learn:
- How does EMI differ from RMI?
- What types of EMI and EMC affect medical devices?
- Shielding options.
- Dealing with computed-tomography interference.
The medical circumstance for many people requires that they have an implanted pacemaker/defibrillator or an implanted insulin pump. These kinds of critical equipment must not be disrupted by the operation of other electronics such as cell phones, etc.
What’s the Difference Between EMI and RFI?
The terms radio-frequency interference (RFI) and electromagnetic interference (EMI) often may be used interchangeably since radio waves are simply a subset of the electromagnetic spectrum. However, there’s actually a difference in practice.
EMI is usually characterized as any frequency of electrical noise; RFI is a specific subset of electrical noise within the EMI spectrum. RFI is classified as a disturbance that may affect an electrical circuit due to either electromagnetic conduction or electromagnetic radiation emitted from some external source.
IEC 60601-1-2 is the primary standard for EMI and electromagnetic compatibility (EMC) in electronic medical equipment and systems. This standard is created to define the safety and performance expectations for medical electronic equipment that is in the presence of an electromagnetic interference.
Medical Device Examples
There are so many types of medical devices that can be worn or implanted. Some of the main examples include:
- Clothing
- Watch
- Ring
- Shoes
- Belt
- Glasses
- Armband
EMI/EMC Effects on Implanted or Wearable Electronics
Electromagnetic waves, which are emitted via communication devices (cell phones, etc.) or from an electrostatic discharge (ESD), will cause power disturbances that create a surge in conductive current as well as a localized radiated transient field.
READ MORE: Why is Industrial Design Important for Wrist-Based Wearables?
EMI may occur when these three factors are present: a source of EMI, a coupling path and a receptor. The coupling path from the source to the receptor can be a magnetic field, electric current or even an electromagnetic field.
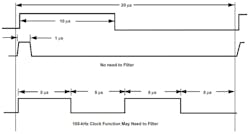
For instance, lightning is an EMI source that occurs in nature. Other sources of EMI might be radios, wireless networks, computers, cellphones or even any electric devices designed to transmit signals. Keep in mind that any signals less than 50 kHz will typically not be a cause of EMI concern (see figure).
Shielding Against EMI
Modern EMI shielding is more challenging than it has ever been before. The use and production of new medical devices, along with the construction of digital circuits that are smaller yet faster for these newer models, make them susceptible to electromagnetic fields and the inevitable interference.
Gold is one of the best coatings that can be employed as an effective shield against EMI. Gold will not cause a reaction when it comes into contact with the human body, Thus, it’s a superior EMI coating for medical devices such as pacemakers. Occasionally, platinum also is used for shielding on pacemakers. Platinum will help resist corrosion, but cost is a problem since it’s higher than the already-expensive gold.
Multi-Cavity EMI Shielding
Designers of medical devices always need to consider EMI shielding to prevent medical equipment failure and stay compliant with federal regulations. One that front, the multi-cavity EMI shield will help solve various problems associated with shielding technologies. This method consists of a lightweight metallized plastic material that’s able to be molded to fit any design shape.
READ MORE: Pacemakers Study Confirms Cell Phone Interference
The metallized plastic material should be attached to the printed circuit board (PCB) after the surface-mount technology (SMT) reflow process. It will allow for easy component inspection or rework. The plastic material also can be easily removed by hand without damaging the board or the soldering. It has excellent shielding capability and is a very simple attachment mechanism as compared to perforated, soldered metal can shields.
Another option is a polymer-based, non-ferromagnetic shield, as it will not disturb MRI imaging.
EMI/EMC Filters
Employing an EMI filter may help with conducted emissions as well as meet immunity and fast transient requirements of radiated emissions. A power line or mains EMI filter can be installed at the power entry point of equipment to prevent electromagnetic noise from exiting or entering the medical implant/device.
Computed-Tomography (CT) Interference with Medical Devices
The Federal Drug Administration (FDA) has been notified of a small number of reports of adverse events that may be associated with CT imaging of implantable and wearable electronic devices (such as insulin pumps, cardiac implantable electronic devices and neurostimulators).
A CT scanner will directly irradiate the circuitry of certain devices (when the device is visible in the resulting CT image) and may cause sufficient electronic interference to affect their function and operation. Although there’s a low probability of causing clinically significant adverse events, it’s still possible.
Interference can be avoided when the medical device isn’t within the primary X-ray beam of the CT scanner. CT is the preferred tomographic imaging technology for patients with implantable or wearable medical devices. CT is safer for patients with devices of unknown magnetic-resonance-imaging (MRI) safety status, too.
Summary
The rise of advanced wireless technologies creates an almost saturation of the spaces that surround us, along with a high level of EMI. This situation can have particularly negative effects on the latest array of worn and implanted medical devices, and it may present a danger to people using them.
Thus, designers need to “err on the side of caution” when designing worn and implanted medical devices. Methods of EMI shielding and filtering must be considered as a key part of these designs.
This article appeared in Electronic Design.
References
Medical applications, Texas Instruments
PCB Design Guidelines For Reduced EMI
EMI’s Potentially Dangerous Impact on Pacemakers
How Does EMI Affect the Human Body and Brain? | Electronic Design
Interference between CT and Electronic Medical Devices | FDA
EMI/EMC Standards for Medical Devices - VSE
5G Networks: Effects On Radiated Emissions And EMI Shielding (com-power.com)
The Importance of EMI Shielding in Medical Devices - SAT Plating
FDA/CDRH Recommendations for EMC/EMI in Healthcare Facilities | FDA
About the Author
Steve Taranovich
Technical Writer, Phoenix Information Communications LLC
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: