What could be more valuable than a face mask that protects you from the SARS-CoV-2 virus? How about a face mask that offers protection and actually detects whether COVID-19 is present in your breath?
As a method of detection, biosensors check several essential boxes when it comes to providing stability and sensitivity relative to traditional sensory methods. Biosensors are for the most part analytical devices that use a recognition element (biochemical receptor) to convert a biological response into an electrical signal. They may be constructed from a host of materials (such as nanoporous metals and carbon nanotubes) and can be tailored for any number of specific biosensing applications. Is it any wonder they’re often touted for being low cost, portable, robust, and for their potential to be tailored to specific biosensing applications?
These high-sensitivity medical devices are not only widely researched and developed, but have proven their utility across sectors, from medical, marine and environmental to food and manufacturing. What follows are five examples that have been published since the start of 2021. While some are underdevelopment, the research shows both purpose and promise for future biosensing applications.
Biosensor-Embedded Face Masks
The button-activated biosensors consist of three different freeze-dried biological reactions that are sequentially activated by the release of water from a reservoir. Once activated, results are available in 90 minutes at levels of accuracy comparable to PCR (polymerase chain reactions) diagnostic tests. The feat of embedding the wearable freeze-dried cell-free (wFDCF) technology into a face mask is the culmination of three years of work and are a stellar practical application.
Beating Back the Coronavirus - COVID-19 detecting face mask from Wyss Institute on Vimeo.
Smart Wound Dressing
Typically, the only way to know whether a bandaged wound is healing is to remove the dressing. That changed when a low-cost, magnesium-based wound dressing was impregnated with nanosensors that glow under UV light to warn patients if an infection starts.
Developed by a team of scientists and engineers at RMIT University in Melbourne, Australia, the dressing uses the properties of magnesium, which is known to be antimicrobial, anti-inflammatory and highly biocompatible.
The research team synthesized the magnesium hydroxide nanosheets—which are 10,000 to 100,000 times thinner than a human hair—and embedded them onto nanofibers. Nanosheets respond to changes in pH; healthy skin is naturally slightly acidic, while infected wounds are moderately alkaline. Under UV light, the nanosheets glow brightly in alkaline environments and fade in acidic conditions, indicating the different pH levels that mark the stages of wound healing, the researchers noted.
The nanosheets can be integrated into standard cotton bandages. Lab tests showed the magnesium hydroxide nanosheets were non-toxic to human cells, while destroying emerging pathogens such as drug-resistant golden staph and Candida auris. The study, published in ACS Applied Materials and Interfaces, is the first to develop fluorescent magnesium hydroxide nanosheets that could contour to the curves of bandage fibers.
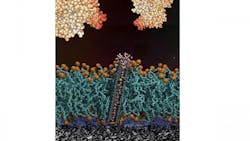
Antifouling Protection
The researchers point out that fouling is a persistent problem that occurs in a four-stage process: First, surfaces immediately become coated with a small layer of molecules. Second, this layer gets covered with the main layer of foulant. Third, the fouled surface begins growing biofilms. Fourth, the biofilm progresses to macrofouling, which usually occurs within days or weeks.
Once formed, biofilms are difficult to remove, said Noy. One example of antifouling protection is a pH sensor with silicon nanowire transistors that are protected by a phospholipid membrane with carbon nanotube pores embedded within the membrane.
Silicon nanowires are efficient pH sensors that provide an electrical signal modulated by solution pH, said Noy. But when they come into contact with a biological medium, they tend to foul up.
The solution was to cover the sensors with a lipid membrane to provide a protein fouling protective barrier. Noy’s research team embedded tiny carbon nanotube pores within the membrane to allow protons to pass through the barrier. The protection allowed the sensors to withstand three-day exposure to protein solutions, milk and blood plasma, while still being able to measure pH, he said.
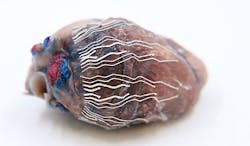
Printable Biosensors Make Images of Organs
A patent-pending 3D-printed biosensor that will soon help surgeons localize critical regions in tissues and organs during operations. Prototypes of the biosensor are under development and can be produced with an automated printing system, noted Chi Hwan Lee, a Purdue University professor of biomedical and mechanical engineering, whose research is published in Nature Communications.
The biosensor material, comprised of sponge-like poroelastic silicone composites, can be adapted for high precision, direct writing of custom-designed stretchable biosensors. Lee described the characteristically soft, thin, stretchable device as being insensitive to strains, enabling it to seamlessly interface with the curvilinear surface of organs. This unique feature enables simultaneous recording and imaging, he said, which facilitates an ability to pinpoint the origin of disease conditions, such as the real-time observations on the propagation of myocardial infarction in 3D.
By using soft bio-inks during rapid prototyping of a custom-fit design, biosensors fit a variety of sizes and shapes of an organ. The researchers tested the prototypes in mice and pigs in vivo. The bio-inks are softer than tissue, stretch without experiencing sensor degradation and have reliable natural adhesion to the wet surface of organs without needing additional adhesives, they said.
Lee and his colleagues also evaluated the biocompatibility and anti-biofouling properties of the biosensors, as well as the effects of the biosensors on cardiac function. They found no significant adverse effects.
Detecting Breast Cancer
A team of researchers at the Polytechnic University of Valencia in Spain have developed, at the laboratory level, a prototype of a biosensor to help detect breast cancer in its earliest stages. The development of this biosensor falls within the field known as liquid biopsy, which helps detect the presence of cancer through a blood test. The mesoporous biosensor developed by the UPV-INCLIVA team is easy to use, low cost and provides results within 30 to 60 minutes from a sample of the patient’s plasma.
The biosensor is composed of a nanomaterial—a nanoporous alumina—that facilitates the detection in plasma of miR-99a-5p microRNA, which is associated with breast cancer, noted researchers. Until now, this has been done by using complex and time-consuming techniques, which means that this system could not be used as a diagnostic tool in the clinical setting.
UPV professor Ramón Martínez Máñez explained that the nanopores of the biosensor are loaded with a dye—rhodamine B—and sealed with an oligonucleotide. When interacting with the plasma sample, if the pore gates do not detect the presence of the microRNA, they remain closed; in contrast, in the presence of miR-99a-5p, the pore gates open and the dye is released. In other words, the change in dye release could mean either healthy or breast cancer patients, said Martínez Máñez.
About the Author

Rehana Begg
Editor-in-Chief, Machine Design
As Machine Design’s content lead, Rehana Begg is tasked with elevating the voice of the design and multi-disciplinary engineer in the face of digital transformation and engineering innovation. Begg has more than 24 years of editorial experience and has spent the past decade in the trenches of industrial manufacturing, focusing on new technologies, manufacturing innovation and business. Her B2B career has taken her from corporate boardrooms to plant floors and underground mining stopes, covering everything from automation & IIoT, robotics, mechanical design and additive manufacturing to plant operations, maintenance, reliability and continuous improvement. Begg holds an MBA, a Master of Journalism degree, and a BA (Hons.) in Political Science. She is committed to lifelong learning and feeds her passion for innovation in publishing, transparent science and clear communication by attending relevant conferences and seminars/workshops.
Follow Rehana Begg via the following social media handles:
X: @rehanabegg
LinkedIn: @rehanabegg and @MachineDesign
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: