Piezoelectric materials are used in everyday applications. Butane lighters, gas grills, cell phones and vibration sensors are among the common appliances and components that draw on piezoelectricity in some way.
Simply defined, piezoelectricity is produced when certain materials generate an electric charge in response to mechanical stress. Several naturally occurring substances demonstrate the piezoelectric effect. Examples include bone, crystals, silk and enamel.
Scientists explore these materials for applicability in biomedical applications, particularly when they display biocompatibility and biodegradability. In medical devices, sensors, actuators and energy harvesting instruments developed from natural or synthetic piezoelectric materials can, for example, convert pressure and flow rate to either consume or generate energy.
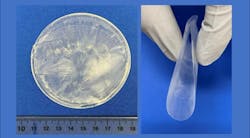
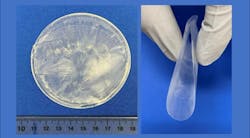
A novel example focuses on implantable piezoelectric therapeutic devices. Bioengineers at the University of Wisconsin’s Department of Materials Science and Engineering are taking advantage of the piezoelectric properties of non-rigid, nontoxic biological materials such as silk, collagen and amino acids.
The researchers explained that the self-assembly process incorporates a biocompatible polymer shell that surrounds the lysine as the polymer/lysine solution evaporates. A chemical interaction occurs between the inner layer of lysine and the polymer coating. This interaction, noted the researchers, is critical for orienting the lysine into the crystal structure necessary for it to produce electric current when flexed.
“This work is an outstanding example of using the chemical properties of the materials to create a self-assembling product,” said David Rampulla, director of the Division of Discovery Science and Technology at the National Institute of Biomedical Imaging and Bioengineering.
The process is rapid and inexpensive, he said, so it adds to the feasibility of using the wafers in therapeutic applications.
“That the wafers are biodegradable opens the possibility for creating electrotherapies that could be used to accelerate healing of an injured bone or muscle, for example, and then degrade and disappear from the body,” said Rampulla.
The research team tested the piezoelectric wafers in rats. Wafers were transplanted into the rats’ legs and chests. The researchers explained that the leg movements and chest movements compressed the piezoelectric wafers enough to create an electrical output.
Blood tests done after the transplanted wafer dissolved in the rats showed normal levels of blood cells and other metabolites. This indicates there were no harmful effects from the dissolved implant, the researchers said.
The work of turning muscle movements into therapeutic approaches can be game-changing, noted Wang. “We believe the technology opens a vast array of possibilities including real-time sensing, accelerated healing of wounds and other types of injuries, and electrical stimulation to treat pain and other neurological disorders,” said Wang. “Importantly, our rapid self-assembling technology dramatically reduces the cost of such devices, which has the potential to greatly expand the use of this very promising form of medical intervention.”
The results were reported in the journal Science.
About the Author

Rehana Begg
Editor-in-Chief, Machine Design
As Machine Design’s content lead, Rehana Begg is tasked with elevating the voice of the design and multi-disciplinary engineer in the face of digital transformation and engineering innovation. Begg has more than 24 years of editorial experience and has spent the past decade in the trenches of industrial manufacturing, focusing on new technologies, manufacturing innovation and business. Her B2B career has taken her from corporate boardrooms to plant floors and underground mining stopes, covering everything from automation & IIoT, robotics, mechanical design and additive manufacturing to plant operations, maintenance, reliability and continuous improvement. Begg holds an MBA, a Master of Journalism degree, and a BA (Hons.) in Political Science. She is committed to lifelong learning and feeds her passion for innovation in publishing, transparent science and clear communication by attending relevant conferences and seminars/workshops.
Follow Rehana Begg via the following social media handles:
X: @rehanabegg
LinkedIn: @rehanabegg and @MachineDesign
Voice Your Opinion!
To join the conversation, and become an exclusive member of Machine Design, create an account today!

Leaders relevant to this article: