|
Authored by: |
Physicians have been taking advantage of technology to help wounds heal for centuries by using bandages, stitches, and more recently, staples and adhesives. And fortunately, most wounds eventually heal themselves, But there are situations in which wounds just won’t heal. These chronic wounds — usually defined as those that show no improvement after 30 days — include bed sores in paralyzed or immobilized people, foot and leg sores on diabetics, and large traumatic wounds due to car accidents or combat.
Fortunately, biomedical engineers and material scientists have developed several new devices and therapies that work on chronic wounds.
Better dressings
One of the simplest new approaches to wound therapy are bioceramic dressings from Cerdak, a South African company. The FDA-approved dressing, called Cerdak, consists of a sachet of loosely packed alumina-oxide spheres ranging in diameter from 0.2 to 1 mm. There are many voids between the nonallergenic spheres and each sphere has many pores. In fact, pores make up about 75% of the spheres’ volume. The pores average about 2 to 5 µm in diameter, which creates capillary suction forces of about 56 kPA when in contact with liquids.
When placed in or near a wound, the spheres attract and retain fluid. As they remove older fluid, the body reacts by supplying more, which contains fresh antigens and the growth hormones and proteins needed to fight infection, grow new tissue and blood vessels, and heal the wound. And studies show the spheres reduce the amount of bacterial toxins and free radicals, which are both inimical to healing.
The standard Cerdak dressing is about the size of a tea bag, but the company makes specialized dressings for wounds in various locations. For example, there are large ones for cavity wounds, specially shaped ones for finger-tip and toe injuries, smaller dressings for facial wounds, and die-cut versions for nonhealing wounds around catheter entry points. The dressings come with and without adhesives.
Negative pressure
Negative-pressure wound therapy (NPWT) involves applying a vacuum to a wound, about –125-mm Hg for up to 22 hr a day, continuous or intermittent, and sealing the wound to retain the vacuum. NPWT has proven effective on acute and chronic wounds, as well as second and third-degree burns. Doctors have to be careful where NPWT is used, but there seems to be no downside to the therapy.
NPWT is thought to cure wounds through five proven mechanisms:
• Suction pulls edges of a wound closer together, reducing the distance needed to be filled by new tissue or even completely closing the wound.
• Stimulates tissue growth that will fill in the wound and replace lost tissue.
• Continually cleans wound, which reduces the bacteria count and chances of infection, and removes substances that inhibit healing.
• Removes pus and other fluids exuded by the wound.
• Reduces swelling and edema, which takes pressure off surrounding capillaries and lets blood flow back into the area.
NPWT also minimizes scarring.
Putting suction on the wound is also thought to mechanically stress the surrounding tissue which could cause the local cells to release chemical agents that promote skin and cell formation.
Firms that make NPWT often differentiate their products by the wound dressings they provide. Kinetic Concepts, San Antonio, for example, uses open-celled reticulated polyurethane foam that evenly distributes the negative pressure across the wound. The foam is covered with a transparent film that keeps bacteria out of the wound. The film also contributes to a tightly sealed vacuum. Other companies add silver or other antibiotics to the foam.
Innovative Therapies, Gaithersburg, Md., uses a foam based on water-blown polyurethane, a hydrophilic or water-loving polymer. According to the company, this foam will not dry out and damage the wound due to lose of flexibility. The company points out that other medical devices use foams made with polyvinyl alcohol (PVA), which must be kept wet if they are to remain flexible.
There are also firms that use gauze with their NPWTs. Health technicians place nonadherent gauze in the wound where it won’t stick to the surface, making it less painful to change dressings. They fill the rest of the wound with moistened gauze. The gauze needs to be covered by an airtight material, usually a film. Health-care workers change the dressings after two days, then two to three times per week.
A third type of dressing, BioDome, was developed at ConvaTec, Skillman, N. J. It is made of layers of nonwoven polyester with a silicone elastomer sandwiched in-between. The surface that touches the wound is studded with hundreds of BioDomes, small, semirigid domes. The domes keep the dressing in contact with the wound but leave space for tissue growth. BioDomes dressing are usually coupled with lower wound pressures, about –75-mm Hg. High pressures can collapse the domes.
The key component in an NPWT dressing is the pump, which can be battery powered and portable, or a bedside device. The pump removes liquids through the tube and usually stores it in a reusable canister. With NPWT, doctors can also irrigate the wound, feeding in cleansing solutions along with vitamins, antibiotics, and other drugs, because the pump continually removes excess fluid. Many NPWT pumps have alarms for over and underpressurization.
Doctors and biomedical engineers are studying various wound-healing techniques on a variety of wounds, both acute and chronic, to assess document savings in time and money, as well as better patient outcomes.
|
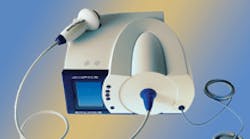
Ultrasound kickstarts healing processes Researchers at Sanuwave Health Inc., Alpharetta, Ga., have discovered a way to restart the healing process and get that cascade flowing again using ultrasound stimulation with a device they design and manufacture called dermaPACE. (PACE stands for pulsed acoustic cellular expression). It uses a handheld transmitter to send four impulses/second of shock-wave energy carrying 0.23 J/mm2. These impulses travel into the tissue or wound where they create shock waves of about 500 bar followed by negative pressure waves of about –90 bar, according to the company. These pressure waves travel about 10 mm into the wound where they generate rapid compression and tension forces at the cellular levels. These forces seem to put mechanical stresses on cells which initiates the healing cascade. In fact, a single treatment, which consists of a little over 2 min of impulse delivery, has been shown to increase the healing response for up to 12 weeks. Patients usually receive four treatments spread over four weeks. The transmitter/applicator contains electronics and liquid components that cannot be sterilized, so health-care technicians encase it in a sterile sleeve while gliding it over the wound to prevent cross contaminations between patients. The sleeve material readily transfers acoustic energy. The technicians also apply a sterile gel on the wound to facilitate the shock waves passing from the applicator to the tissue. Although dermaPACE and similar devices have been shown to initiate and accelerate healing burns, traumatic wounds, reconstructive skin grafts, and diabetic wounds, Sanuwave is initially targeting the device for use on foot ulcers of diabetics, one of the most common chronic wounds. One of the symptoms of diabetes is neuropathy, nerve damage that leads to a loss of feeling. So diabetic patients in clinical tests and trials have not complained of pain. And patients who were treated for other types of wounds did not report pain either, pperhaps becuase the shock waves do not penetrate far intop the body. But burns studies from Europe report they do use lidocain, a local anesthetic, just prior to dermaPACE treatments. The FDA is currently reviewing clinical trials of the device for use on diabetic sores. It is evaluating time to wound closure, reduction in wound size, rate of patient improvement, patient long-term safety, and skin appearance and pain reduction. If all goes well, the agency could approve the device relatively soon. And the AMA is already looking favorably on the device based on published clinical data and support from several medical societies. |
|
High-tech therapy for Third‑World countries One of the more practical advantages in this instance was that, if successful, the device would reduce the need to change wound dressings, a painful experience, from three times per day to once every few says. There are portable NPWT machines, but they weigh 5 to 10 lb, require a power source or lots of batteries to run the pump, and can cost up to $100 a day to rent. Zurovvcik’s half-pound device consists of a simple bellows pump and tube and an airtight adhesive plastic bandage. During development, Zurovcik tweaked the bellows so that the power needed to keep a vacuum beneath the bandage dropped from 14 W to 80 mW. The unit was used on eight patients with wounds ranging from amputations to relatively simple bedsores. The team recruited family members of the patients to do nursing duties, including keeping the vacuum up by periodically pumping the bellows. Unfortunately, the team could only stay in the country for 10 days, not long enough to fully evaluate the treatment and the device. But doctors say the device seemed to keep the wounds cleaner and definitely cut down on bandage changes. In fact, one injured man in the bed next to a person participating in the study requested the treatment when he saw that it led to fewer bandage changes. |